
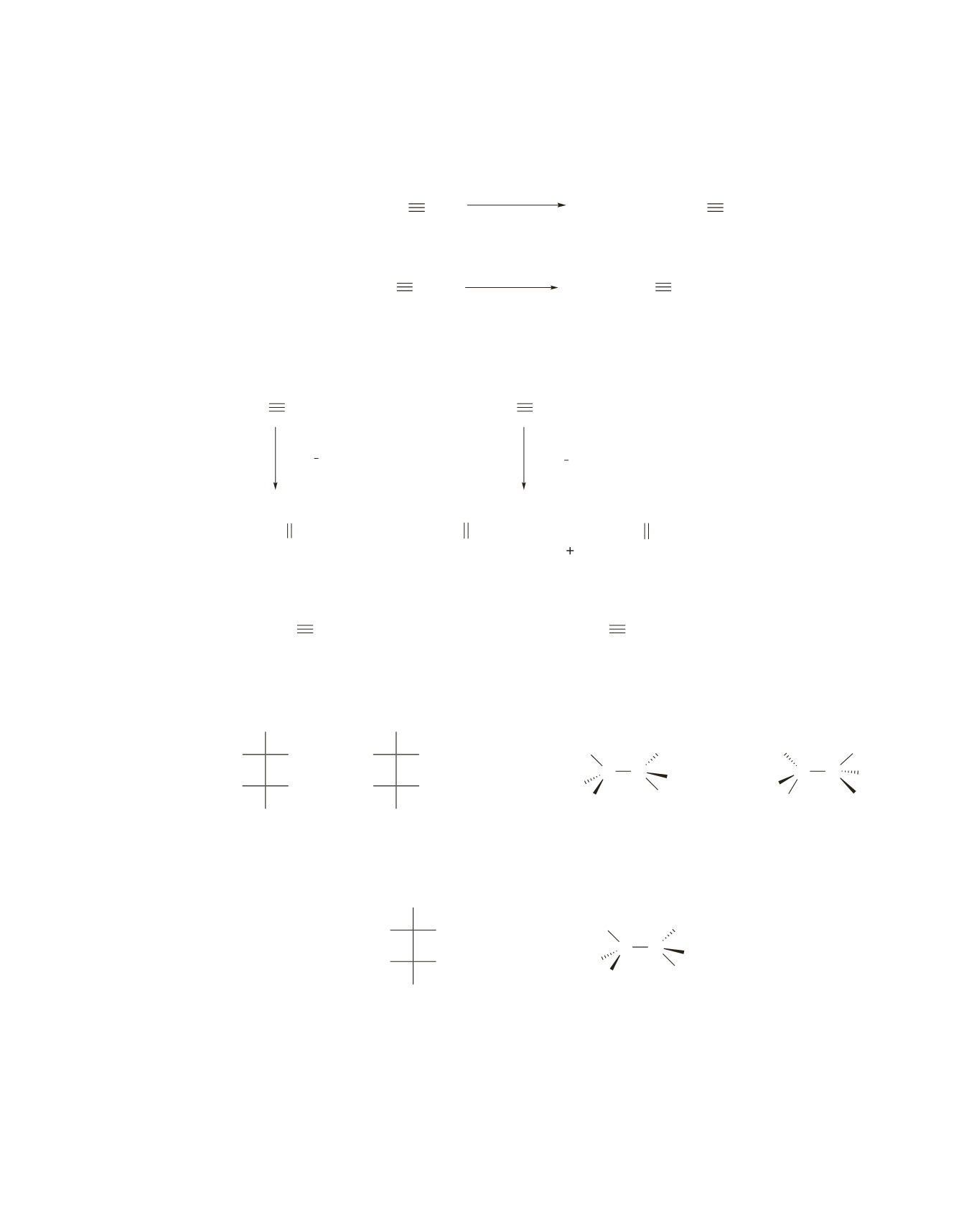
Chapter 7 255
Copyright © 2017 Pearson Education, Inc.
53.
The chemist can make 3-octyne by using 1-hexyne instead of 1-butyne. He would then need to use ethyl
bromide (instead of butyl bromide) for the alkylation step:
CH
3
CH
2
CH
2
CH
2
C CH
1. NaNH
2
2. CH
3
CH
2
Br
CH
3
CH
2
CH
2
CH
2
C CCH
2
CH
3
Or he could make the 1-butyne he needed by alkylating ethyne:
1. NaNH
2
2. CH
3
CH
2
Br
HC CH
CH
3
CH
2
C CH
54.
a.
Only one product is obtained from hydroboration–oxidation of 2-butyne because the alkyne is
symmetrical. Two different products can be obtained from hydroboration–oxidation of 2-pentyne
because the alkyne is not symmetrical.
CH
3
CH
2
CCH
3
CH
3
C CCH
3
CH
3
C CCH
2
CH
3
CH
3
CCH
2
CH
3
CH
3
CH
3
CH
2
CCH
2
CH
3
1. R
2
BH/THF
1. R
2
BH/THF
2. HO , H
2
O
2
, H
2
O
2. HO , H
2
O
2
, H
2
O
O
O
O
b.
Only one product is obtained from hydroboration–oxidation of a symmetrical alkyne such as 3-hexyne
or 4-octyne.
CH
3
CH
2
C CCH
2
CH
3
CH
3
CH
2
CH
2
C CCH
2
CH
2
CH
3
3-hexyne
4-octyne
55.
a.
The first step forms a trans alkene. Syn addition to a trans alkene forms the threo pair of enantiomers.
CH
2
CH
3
H D
CH
2
CH
3
D H
CH
2
CH
3
D H
CH
2
CH
3
H D
+
or
CH
3
CH
2
CH
2
CH
3
H
H
D
D
C C
H
D
CH
2
CH
3
D
CH
3
CH
2
H C C
b.
The first step forms a cis alkene. Syn addition to a cis alkene forms the erythro pair of enantiomers, but
because each asymmetric carbon is bonded to the same four groups, the product is a meso compound.
CH
2
CH
3
H D
CH
2
CH
3
H D
or
CH
3
CH
2
CH
2
CH
3
D
H
D
H
C C
















