
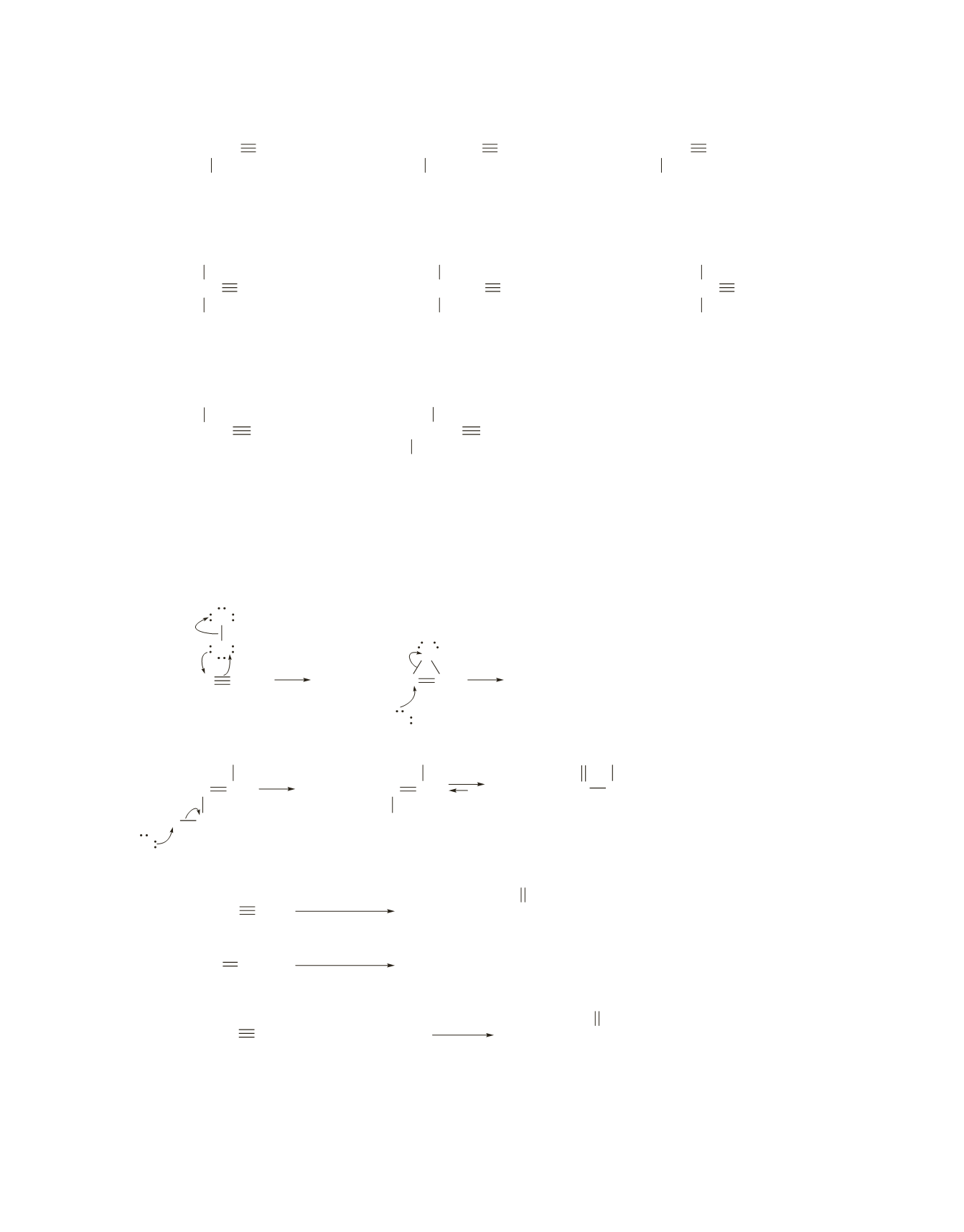
250 Chapter 7
Copyright © 2017 Pearson Education, Inc.
CH
3
CH
2
CHC CCH
3
4-methyl-2-hexyne
sec
-butylmethylacetylene
CH
3
CH
3
CHCH
2
C CCH
3
5-methyl-2-hexyne
isobutylmethylacetylene
CH
3
CH
3
CHC CCH
2
CH
3
2-methyl-3-hexyne
ethylisopropylacetylene
CH
3
*
CH
3
CC CCH
3
4,4-dimethyl-2-pentyne
tert
-butylmethylacetylene
CH
3
CH
3
CH
3
CCH
2
C CH
4,4-dimethyl-1-pentyne
CH
3
CH
3
CH
3
CH
2
CC CH
3,3-dimethyl-1-pentyne
tert
-pentylacetylene
CH
3
CH
3
CH
3
CHCHC CH
CH
3
CH
3
3,4-dimethyl-1-pentyne
CH
3
CH
2
CHC CH
CH
2
CH
3
3-ethyl-1-pentyne
*
b.
There are 14 alkynes if stereoisomers are ignored. Four of the 14 alkynes have an asymmetric center,
so each of these can have either the
R
or
S
configuration. Therefore, if stereoisomers are included, there
would be 18 alkynes.
36.
37.
a.
CH
3
CH
2
CH
2
C CH
CH
3
CH
2
CH
2
CH
2
CH
O
1. R
2
BH/THF
2. HO, H
2
O
2
–
CH
3
CH
2
CH CH
2
CH
3
CH
2
CH
2
CH
2
OH
H
2
O ,
1. R
2
BH/THF
2. HO, H
2
O
2
–
H
2
O ,
b.
CH
3
CH
2
CH
2
C CH
CH
3
CH
2
CH
2
CH
2
CH
O
1. R
2
BH/THF
2. HO, H
2
O
2
–
CH
3
CH
2
CH CH
2
CH
3
CH
2
CH
2
CH
2
OH
H
2
O ,
1. R
2
BH/THF
2. HO, H
2
O
2
–
H
2
O ,
c.
CH
3
CH
2
CH
2
C CCH
2
CH
2
CH
3
CH
3
CH
2
CH
2
CCH
2
CH
2
CH
2
CH
3
O
H
2
SO
4
H
2
O
+
This symmetrical alkyne will give the greatest yield of the desired ketone.
Because the reactant is not a terminal alkyne, the reaction can take place without the mercuric ion
catalyst.
CH
3
CH
2
CH
2
C CCH
Br
Br
Br
H
2
O
H
H
2
O
CH
CH
3
CH
2
CH
2
C
Br
OH
CH
CH
3
CH
2
CH
2
C
Br
OH
CH
CH
3
CH
2
CH
2
C
enol
O Br
CH
CH
3
CH
2
CH
2
C
+
















