
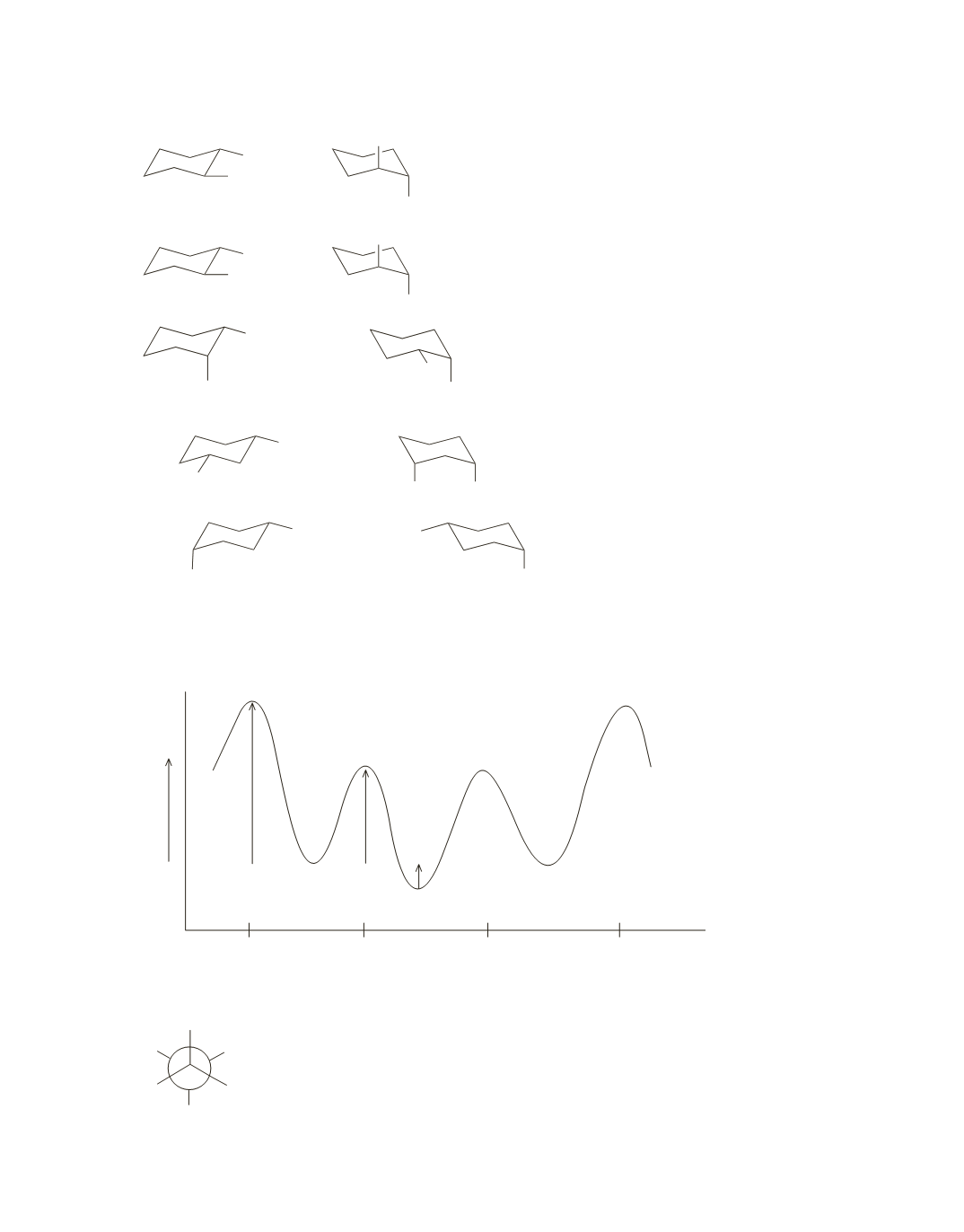
152 Chapter 3
Copyright © 2017 Pearson Education, Inc.
b.
CH
2
CH
3
more stable
CH
2
CH
3
CH(CH
3
)
2
CH(CH
3
)
2
c.
CH
2
CH
3
more stable
CH
2
CH
3
CH
3
CH
3
d.
CH
2
CH
3
CH
2
CH
3
CH
2
CH
3
CH
3
CH
2
equally stable
e.
CH
2
CH
3
CH
2
CH
3
(CH
3
)
2
CH
(CH
3
)
2
CH
more stable
f.
CH
2
CH
3
CH
2
CH
3
(CH
3
)
2
CH
more stable
(CH
3
)
2
CH
75.
Alcohols with low molecular weights are more water soluble than alcohols with high molecular weights
because, with fewer carbons, they have a smaller nonpolar component that has to be dragged into water.
76.
a.
Potential Energy
Dihedral Angle
0º
120º
240º
360º
9.3
kcal/mol
5.2
kcal/
mol
1.2
kcal/mol
b.
H
Cl
H
Cl
H
H
c.
1.2
+
5.2
=
6.4 kcal
>
mol
d.
1.2
+
9.3
=
10.5 kcal
>
mol
















