
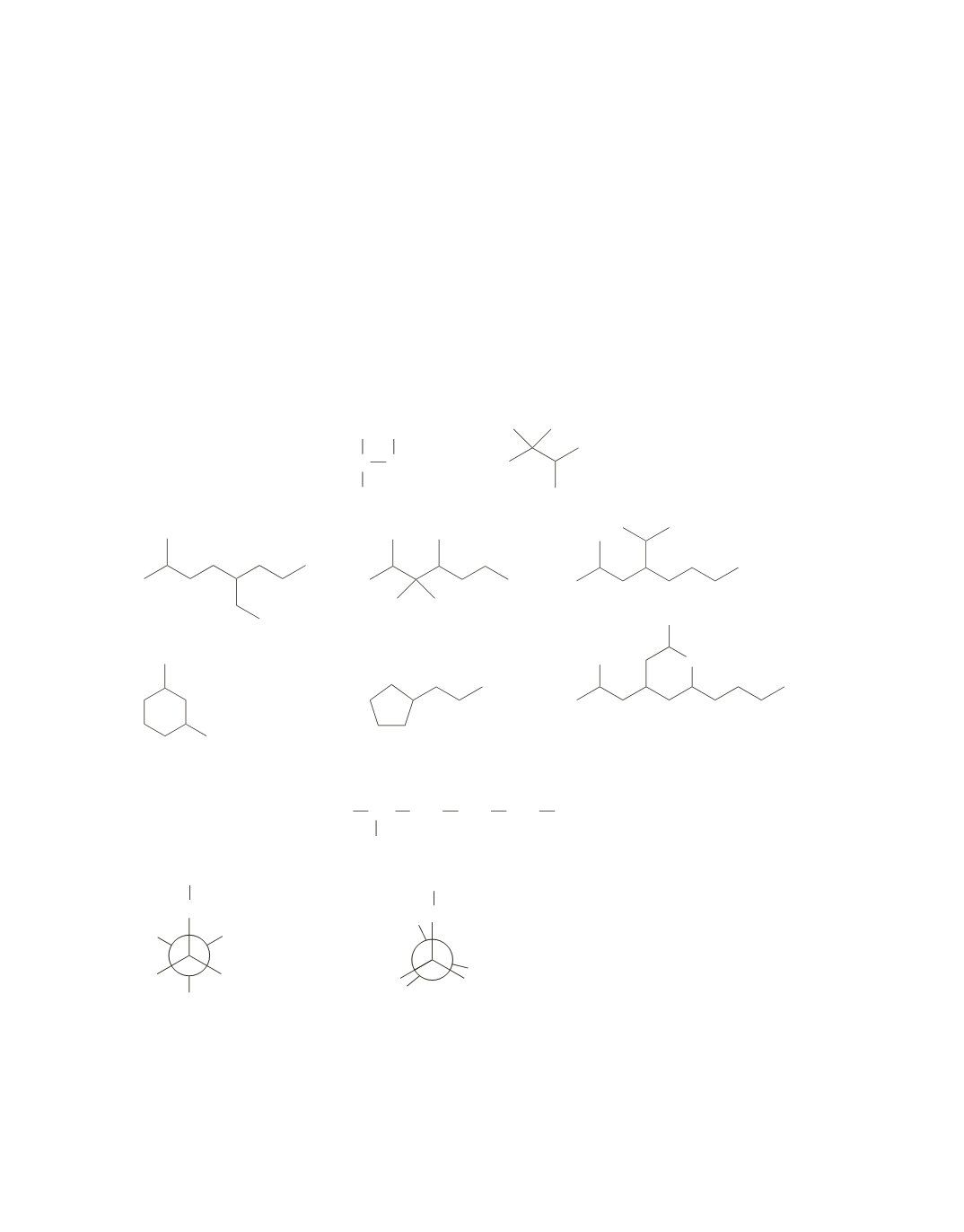
150 Chapter 3
Copyright © 2017 Pearson Education, Inc.
67.
The student named only one compound correctly.
a.
2-bromo-3-pentanol
b.
4-ethyl-2,2-dimethylheptane
c.
3-methylcyclohexanol
d.
2,2-dimethylcyclohexanol
e.
5-(2-methylpropyl)nonane
f.
1-bromo-3-methylbutane
g.
correct
h.
2,5-dimethylheptane
i.
5-bromo-2-pentanol
j.
3-ethyl-2-methyloctane
k.
2,3,3-trimethyloctane
l.
N,N
,5-trimethyl-3-hexanamine
68.
All three compounds are diaxial-substituted cyclohexanes.
B
has the highest energy. Only
B
has a
1,3-diaxial interaction between
CH
3
and Cl, which will be greater than a 1,3-diaxial interaction between
CH
3
and H or between Cl and H.
69.
The only one is 2,2,3-trimethylbutane.
CH
3
C CHCH
3
CH
3
CH
3
CH
3
70.
a.
c.
e.
b.
d.
f.
71.
First draw the structure so that you know what groups to put on the bonds in the Newman projections.
CH
3
CH
CH
3
3
4
CH
2
CH
2
CH
2
CH
3
a.
CH
2
CH
3
H
H
CHCH
3
H
H
CH
3
most stable
c.
Rotation can occur about all the C—C bonds. There are six carbon–carbon bonds in the compound,
so there are five other carbon–carbon bonds, in addition to the
C
3
¬
C
4
bond, about which rotation
can occur.
b.
H
CH
3
CH
2
H
CHCH
3
H
H
CH
3
least stable
















