
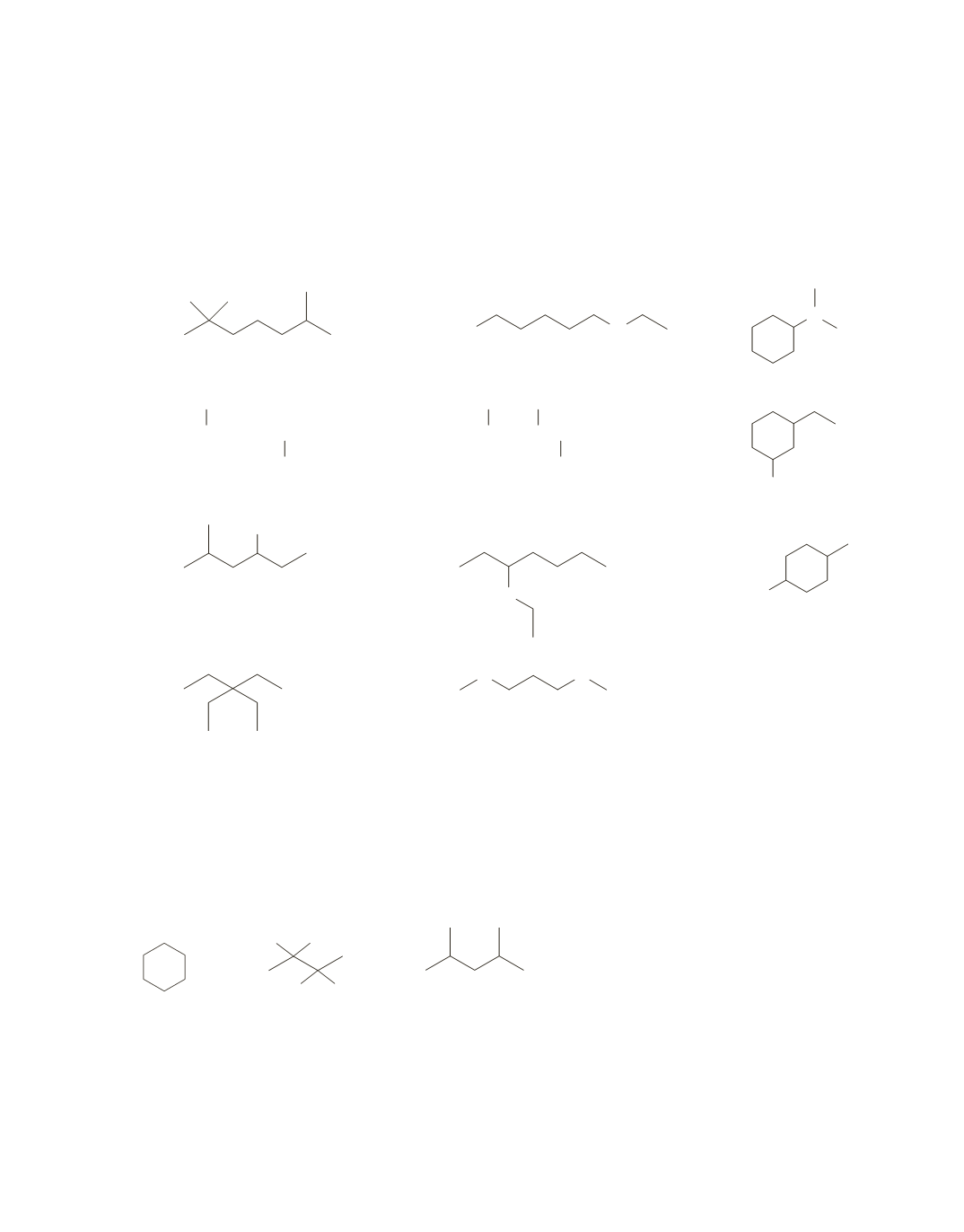
148 Chapter 3
Copyright © 2017 Pearson Education, Inc.
57.
a.
1.
2,2,6-trimethylheptane
2.
5-bromo-2-methyloctane
3.
5-methyl-3-hexanol
4.
3,3-diethylpentane
5.
5-bromo-
N
-ethyl-1-pentanamine
6.
2,3,5-trimethylhexane
7.
3-ethoxyheptane
8.
1,3-dimethoxypropane
9.
N,N-
dimethylcyclohexanamine
10.
3-ethylcyclohexanol
11.
1-bromo-4-methylcyclohexane
b.
2.
1.
5.
3.
6.
7.
OH
9.
10.
11.
4.
Br
N
H
O
N
OH
Br
O
8.
O
CH
3
CHCH
2
CH
2
CHCH
2
CH
2
CH
3
CH
3
Br
CH
3
CHCH
2
CHCHCH
3
CH
3
CH
3
CH
3
58.
C
and
D
are cis isomers. (Both substituents are downward pointing in
C
; both substituents are upward
pointing in
D
.)
59.
a. 1.
3
2.
4
b.
1.
6
2.
5
c. 1.
3
2.
4
60.
The first conformer (
A
) is the most stable because the three substituents are more spread out, so its gauche
interactions will not be as large. (The Cl in
A
is between a
CH
3
and an H, whereas the Cl in
B
and
C
is
between two
CH
3
groups.)
61.
a.
b.
c.
















