
144 Chapter 3
Copyright © 2017 Pearson Education, Inc.
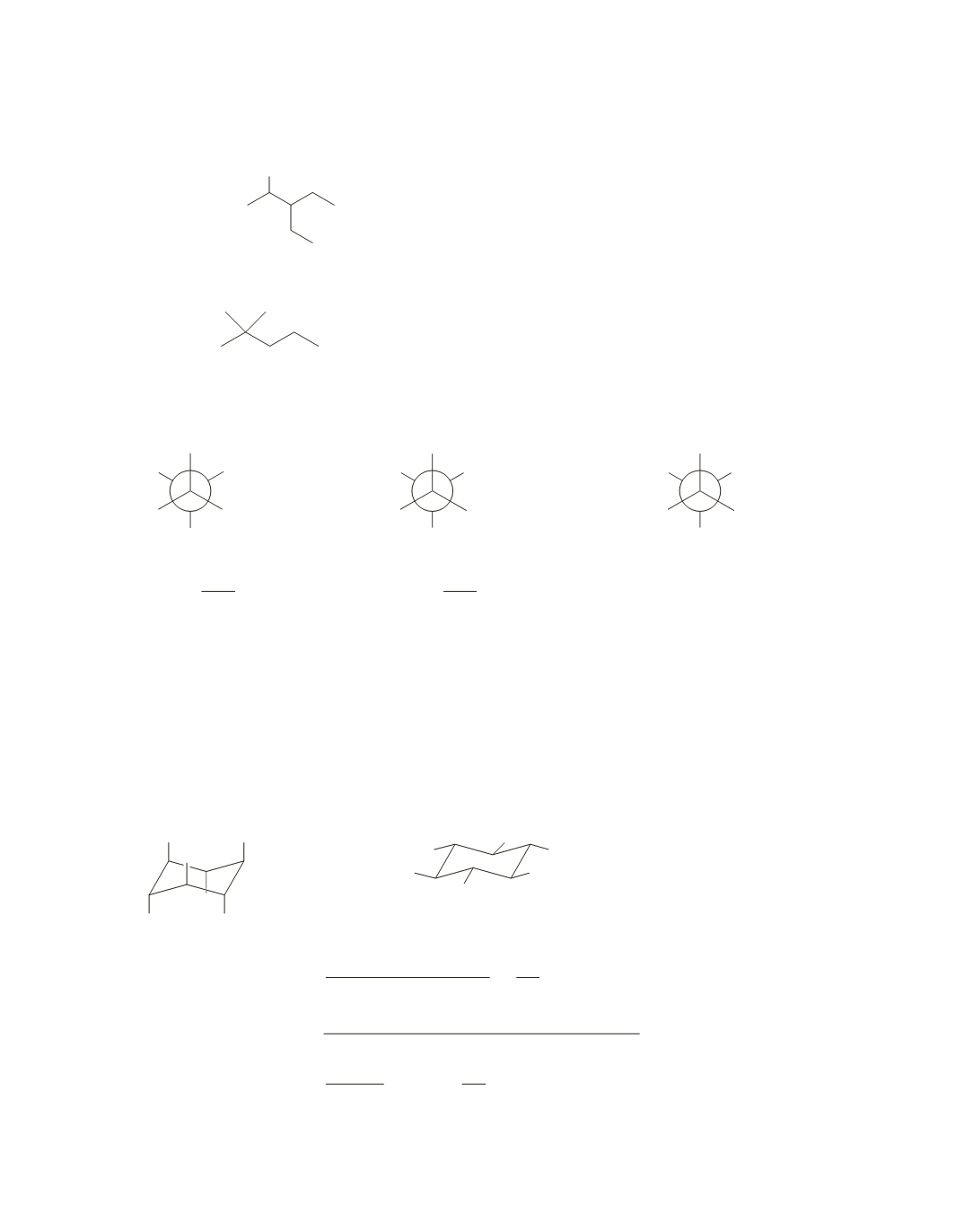
41.
a.
The Newman projection shows rotation about the C-2—C-3 bond.
OH
2
3
3-ethyl-2-pentanol
b.
The Newman projection shows rotation about the C-2—C-3 bond.
2
3
NH
2
2-methyl-2-pentanamine
42.
To draw the most stable conformer: put the largest group on the front carbon opposite the largest group on
the back carbon.
a.
CH
2
CH
3
H
CH
3
CH
3
H
H
b.
CH
2
CH
3
H
H
CH
2
CH
3
CH
3
H
c.
CH
2
CH
3
3
H
H
CH
2
CH
3
CH
3
CH
43.
a.
180
°
-
360
°
8
b.
180
°
-
360
°
9
180
°
-
45
°
=
135
°
180
°
-
40
°
=
140
°
44.
You can get the total strain energy of cycloheptane by subtracting the strainless heat of formation from the
actual heat of formation:
The “strainless” heat of formation of cycloheptane is
7
1
-
4.92
2
=
-
34.4 kcal
>
mol.
The actual heat of formation of cycloheptane is
-
28.2 kcal
>
mol
(from Table 3.8 on page 124 of the text).
Therefore, the total strain energy of cycloheptane is
-
28.2
-
1
-
34.4
2
=
6.2 kcal
>
mol.
45.
a.
Cl
Cl
Cl
Cl
Cl
Cl
b.
Cl
Cl
Cl
Cl
Cl
Cl
46.
K
eq
=
3
equatorial conformer
4
3
axial conformer
4
=
5.4
1
% of equatorial conformer
=
3
equatorial conformer
4
3
equatorial conformer
4
+
3
axial conformer
4
*
100
=
5.4
5.4
+
1
*
100
=
5.4
6.4
*
100
=
84%
















