
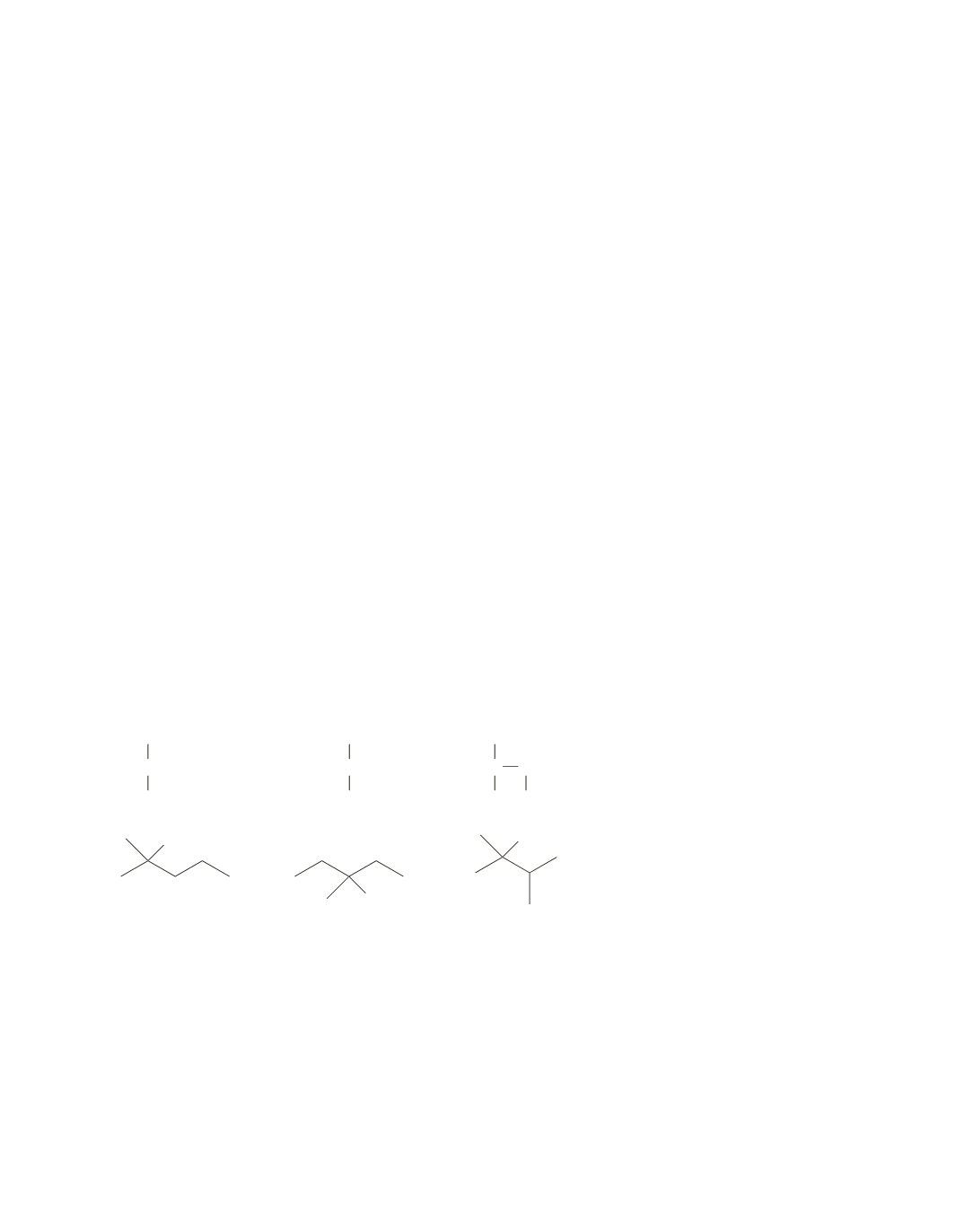
140 Chapter 3
Copyright © 2017 Pearson Education, Inc.
24.
CH
3
OH
common
=
methyl alcohol
systematic
=
methanol
CH
3
CH
2
OH
common
=
ethyl alcohol
systematic
=
ethanol
CH
3
CH
2
CH
2
OH
common
=
propyl alcohol
or
n
-propyl alcohol
systematic
=
1
@
propanol
22.
a. 1.
methoxyethane
2.
ethoxyethane
3.
4-methoxyoctane
4.
1-isopropoxy-3-methylbutane
5.
1-propoxybutane
6.
2-isopropoxyhexane
b.
No.
c. 1.
ethyl methyl ether
2.
diethyl ether
3.
no common name
4.
isopentyl isopropyl ether
5.
butyl propyl ether
6.
no common name
23.
a.
1-pentanol
primary
b.
5-chloro-2-methyl-2-pentanol
tertiary
c.
5-methyl-3-hexanol
secondary
d.
7-methyl-3,5-octanediol (Notice that because there
are two OH groups, the suffix is “diol.”)
both alcohol groups are secondary
CH
3
CH
2
CH
2
CH
2
OH
common
=
butyl alcohol or
n
-butyl alcohol
systematic
=
1
@
butanol
CH
3
CH
2
CH
2
CH
2
CH
2
OH
common
=
pentyl alcohol or
n
-pentyl alcohol
systematic
=
1
@
pentanol
CH
3
CH
2
CH
2
CH
2
CH
2
CH
2
OH
common
=
hexyl alcohol
or
n
-hexyl alcohol
systematic
=
1
@
hexanol
25.
CH
3
CCH
2
CH
2
CH
3
OH
CH
3
OH
CH
3
CH
2
CCH
2
CH
3
OH
OH
CH
3
CH
3
C
OH
CHCH
3
CH
3
CH
3
OH
2-methyl-2-pentanol
3-methyl-3-pentanol
2,3-dimethyl-2-butanol
26.
a.
4-chloro-3-ethylcyclohexanol
secondary
b.
7,8-dimethyl-3-nonanol
secondary
c.
1-bromo-5,5-dimethyl-3-heptanol
secondary
d.
4-methylcyclohexanol
secondary
















