
142 Chapter 3
Copyright © 2017 Pearson Education, Inc.
32.
To be a liquid at room temperature, the compound must have a boiling point that is greater than room
temperature.
pentane or 2-methylbutane
33.
a.
1, 4, and 5
b.
1, 2, 4, 5, and 6
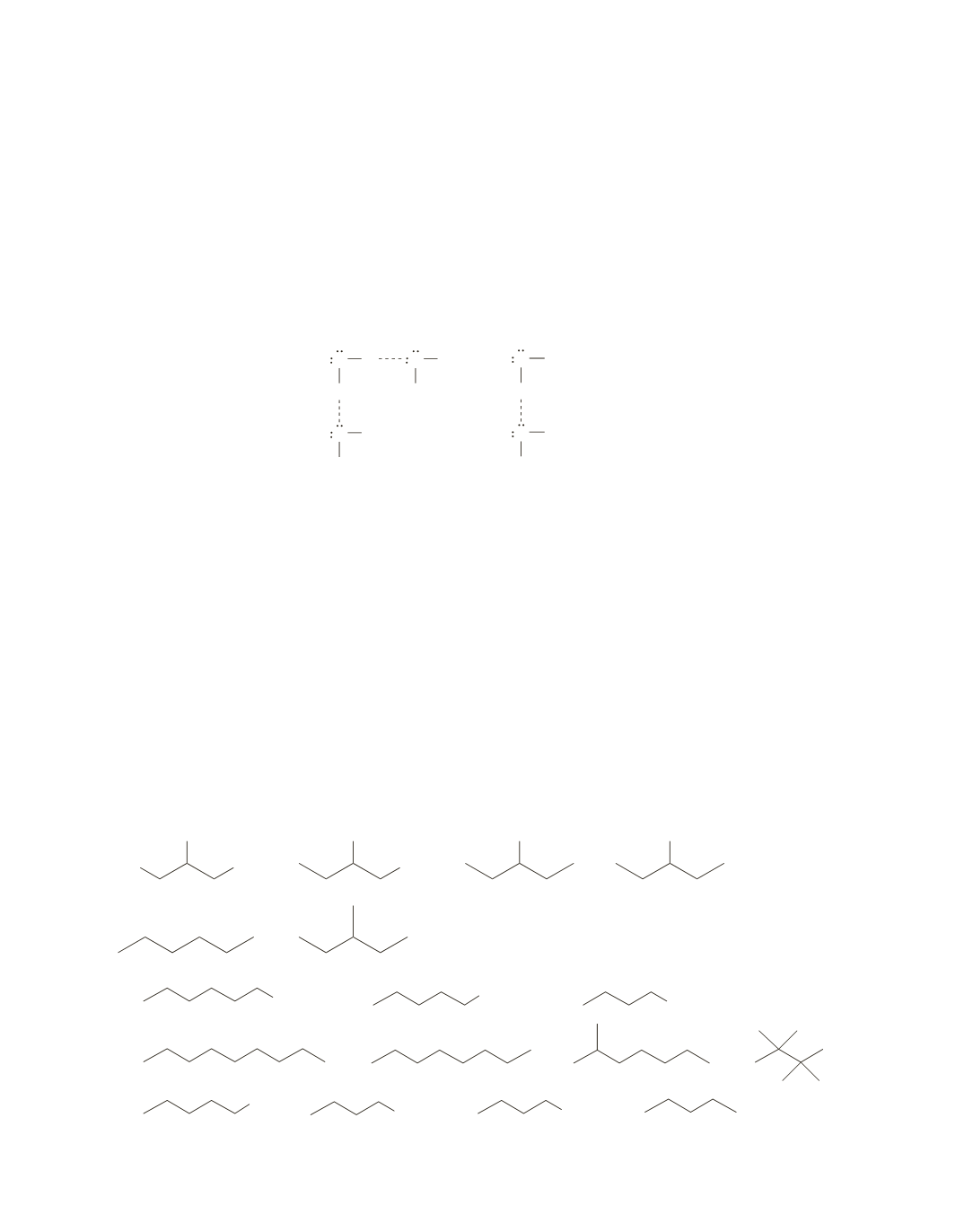
34.
a.
Each water molecule has two hydrogens that can form hydrogen bonds, whereas each alcohol mole-
cule has only one hydrogen that can form a hydrogen bond. Therefore, there are more hydrogen bonds
between water molecules than between alcohol molecules.
O H
H
O H
H
O H
H
O CH
3
H
O CH
3
H
b.
Each water molecule has two hydrogens that can form hydrogen bonds and two lone pairs that can
accept hydrogen bonds.
Ammonia has three hydrogens that can form hydrogen bonds but only one lone pair that can accept
hydrogen bonds.
Therefore, only one hydrogen of an ammonia molecule can engage in hydrogen bonding, so it will
have a lower boiling point than water.
c.
Each water molecule has two hydrogens that can form hydrogen bonds and two lone pairs that can
accept hydrogen bonds.
HF has three lone pairs that can accept hydrogen bonds but only one hydrogen that can form a
hydrogen bond.
Therefore, only one lone pair of a HF molecule can engage in hydrogen bonding, so it will have a
lower boiling point than water.
d.
HF and ammonia can each form only one hydrogen bond, but HF has a higher boiling point because
the hydrogen bond formed by HF is stronger since fluorine is more electronegative than nitrogen.
35.
HO
OH
OH
OH
OH
OH
NH
2
>
>
>
>
>
36.
a.
Br
Br
Br
>
>
b.
>
>
>
c.
OH
OH
Cl
>
>
>
















