
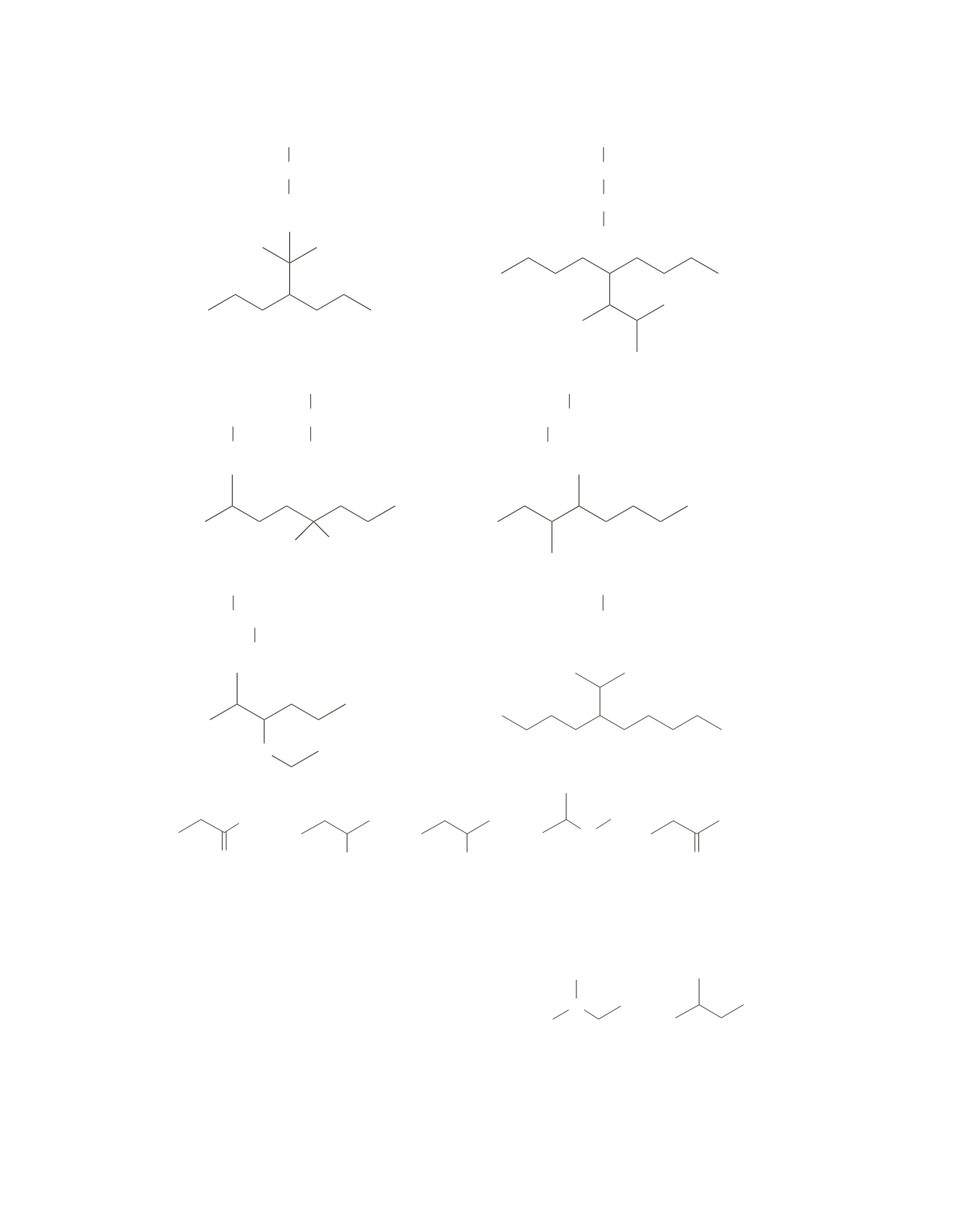
Chapter 3 147
Copyright © 2017 Pearson Education, Inc.
g.
CH
3
CH
2
CH
2
CHCH
2
CH
2
CH
3
CH
3
CCH
3
CH
3
j.
CH
3
CH
2
CH
2
CH
2
CHCH
2
CH
2
CH
2
CH
3
CHCH
3
CHCH
3
CH
3
h.
CH
3
CHCH
2
CH
2
CCH
2
CH
2
CH
3
CH
3
Br
Br
Br
Br
k.
CH
3
CH
2
CHCHCH
2
CH
2
CH
2
CH
3
CH
3
CH
3
i.
CH
3
CHCHCH
2
CH
2
CH
3
O
CH
3
OCH
2
CH
3
l.
CH
2
CH
3
CH
2
CH
2
CHCH
2
CH
2
CH
2
CH
2
CH
3
CHCH
3
CH
3
56.
OH
>
OH
O
NH
2
N
H
O
N
>
>
>
>
>
has two groups that
form hydrogen
bonds
O is more
electronegative
than N, so OH
hydrogen bonds
are stronger than
NH hydrogen
bonds
primary amines
form stronger
hydrogen bonds
than do secondary
amines
no hydrogen bonds;
weaker dipole–dipole
interactions than
oxygen-containing
compounds because
N is less electro-
negative than O
no dipole–dipole
interactions
no hydrogen bonds;
only dipole–dipole
interactions
relatively weak
hydrogen bonds
















