
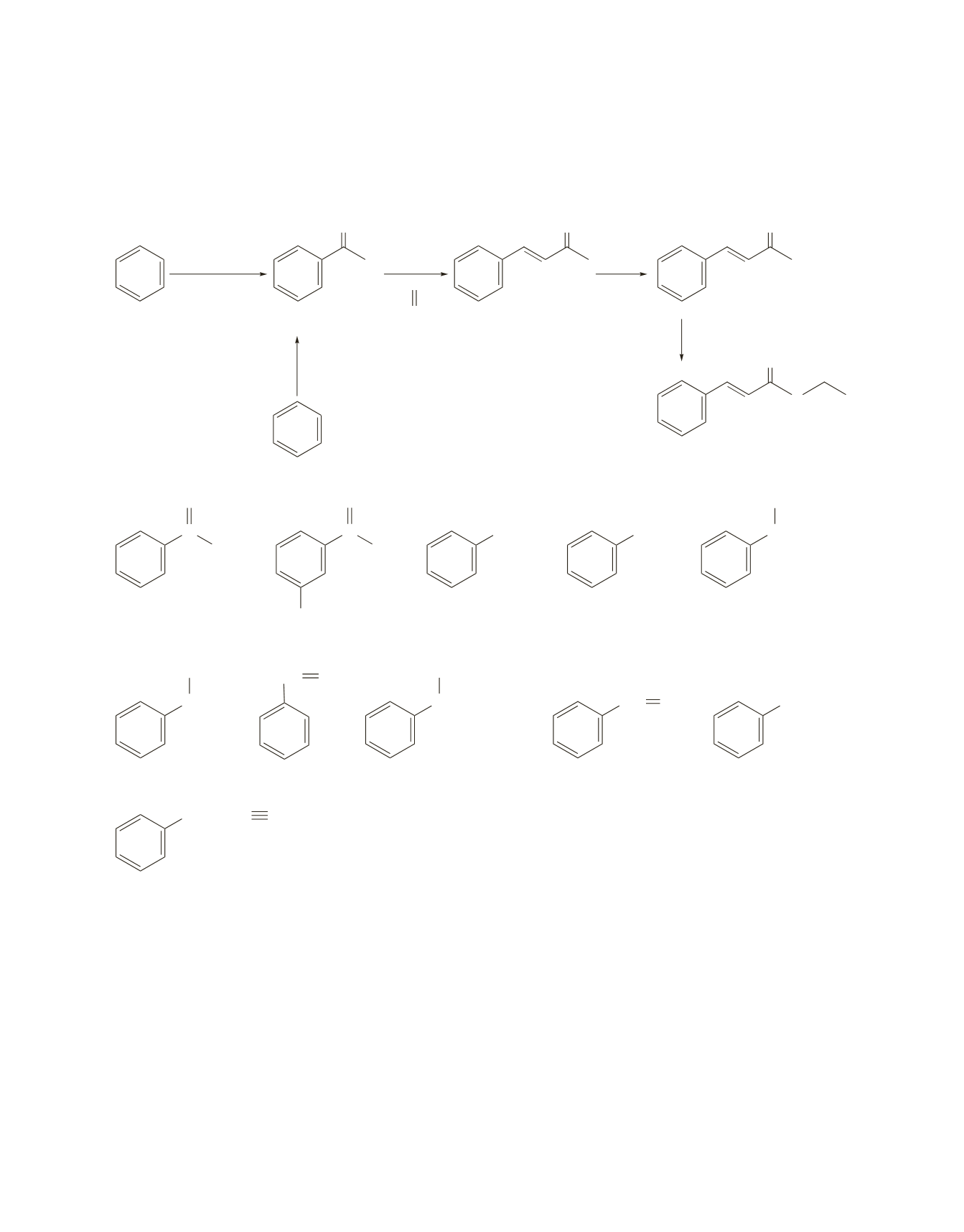
636 Chapter 18
Copyright © 2017 Pearson Education, Inc.
84.
Synthesizing benzaldehyde from benzene would be easy if formyl chloride could be used. However, this
compound is unstable and must be generated in situ via a Gatterman–Koch reaction (see page 884 in the
text), or it can be synthesized via a Friedel–Crafts alkylation reaction. Conversion of the methyl ketone to a
carboxylic acid is called a haloform reaction. (See Chapter 17, Problem 58.)
H
O
O
O
O
−
O
O
1. CH
3
Cl/AlCl
3
2. NBS/peroxide
3. HO
−
4. NaOCl/CH
3
COOH/0
°
C
HO
excess
excess
−
1. HO
−
I
2
O
+
H
2
O
1. SO
2
Cl 2. CH
3
CH
2
OH
AlCl
3
, CuCl
high pressure
CO,
or
Cl
2.
CH
3
CCH
3
add slowly
85.
CH
3
C
O
A
CH
3
C
O
NO
2
CH
2
CH
3
C
B
COOH
D
CHCH
3
E
Br
CHCH
3
CH
H
G
CH
2
CH
2
Br
I
OCH
3
CHCH
3
I
+
CH
3
OH
CH
2
CH
2
CH
2
C
J
N
CH CH
2
F
+
















