
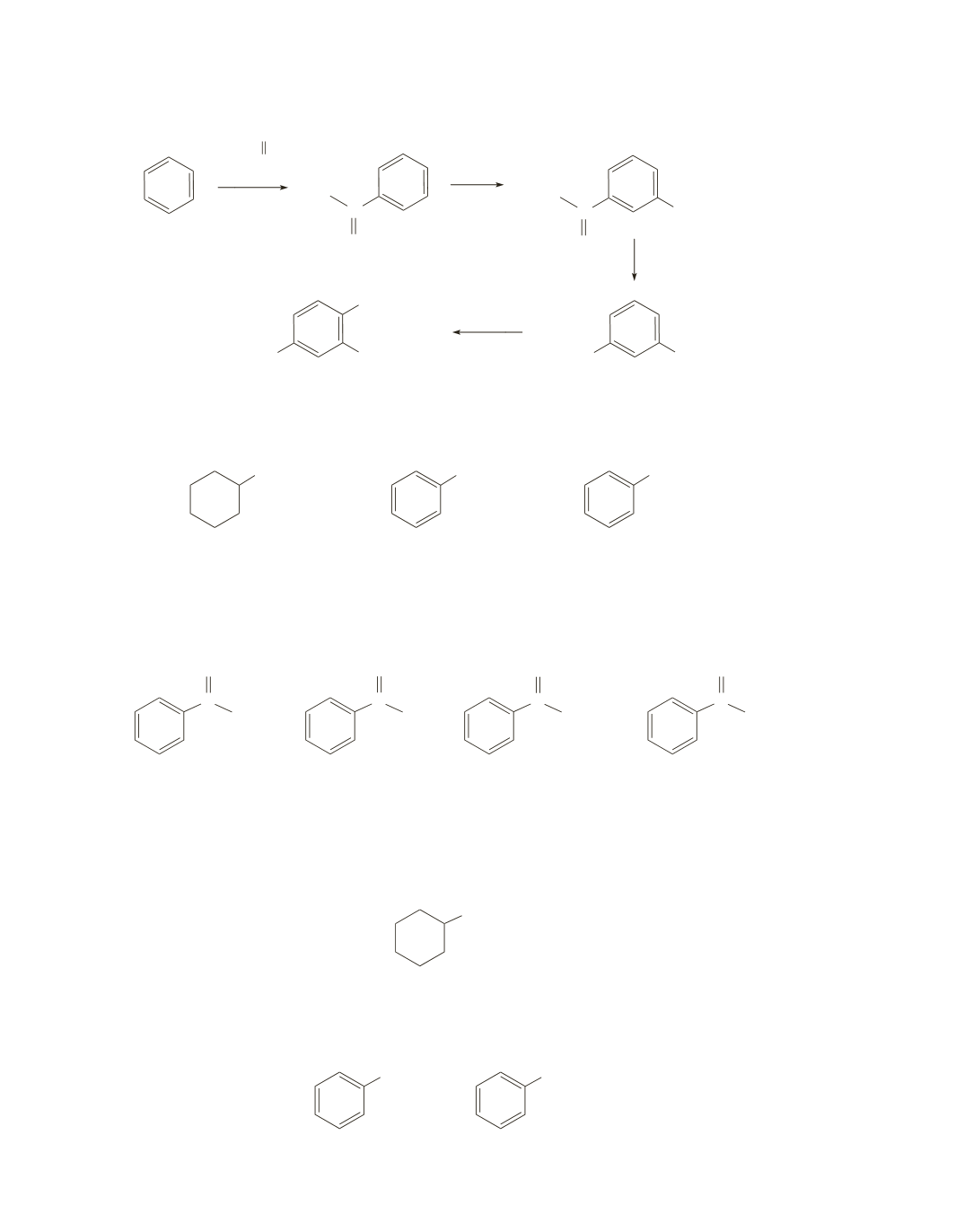
640 Chapter 18
Copyright © 2017 Pearson Education, Inc.
d.
2. H
2
O
Br
2
FeBr
3
C
CH
3
O
Br
CH
3
CH
2
Br
H
2
Pd/C
(CH
3
)
3
CCl
CH
3
CH
2
Br
C(CH
3
)
3
AlCl
3
1. CH
3
CCl
O
AlCl
3
C
CH
3
O
92.
a.
The first three compounds will not show a carbonyl stretch at 1700 cm
-
1
, and the bottom four will
show this absorption band. The first three can be distinguished from one another by the presence or
absence of the indicated absorption bands.
CH
2
OH
CH
2
OH
CH
2
OCH
3
band at 3300 cm
-
1
band at 3300 cm
-
1
no band at 3300 cm
-
1
no band at 1600 cm
-
1
band at 1600 cm
-
1
band at 1600 cm
-
1
The last four compounds all have an absorption band at 1700 cm
-
1
.
They can be distinguished by the presence or absence of the indicated absorption bands.
C
OH
O
C
H
O
C
OCH
O
3
C
CH
O
3
large broad band at
no band at
no band at
no band at
2500
9
3500 cm
-
1
2500
9
3500 cm
-
1
2500
9
3500 cm
-
1
2500
9
3500 cm
-
1
band at 1250 cm
-
1
no band at 1250 cm
-
1
band at 1250 cm
-
1
no band at 1250 cm
-
1
no band at 2700 cm
-
1
band at 2700 cm
-
1
no band at 2700 cm
-
1
no band at 2700 cm
-
1
b.
This is the only compound without the characteristic benzene ring hydrogens at 7
-
8 ppm.
CH
2
OH
Only two compounds will have two signals other than the signals for the benzene ring hydrogens. They can
be distinguished by integration (2:3 versus 2:1) or by the two sharp singlets for the ether versus the some-
what broader singlet for the hydrogen bonded to oxygen.
CH
2
3
OCH
CH
2
OH
















