
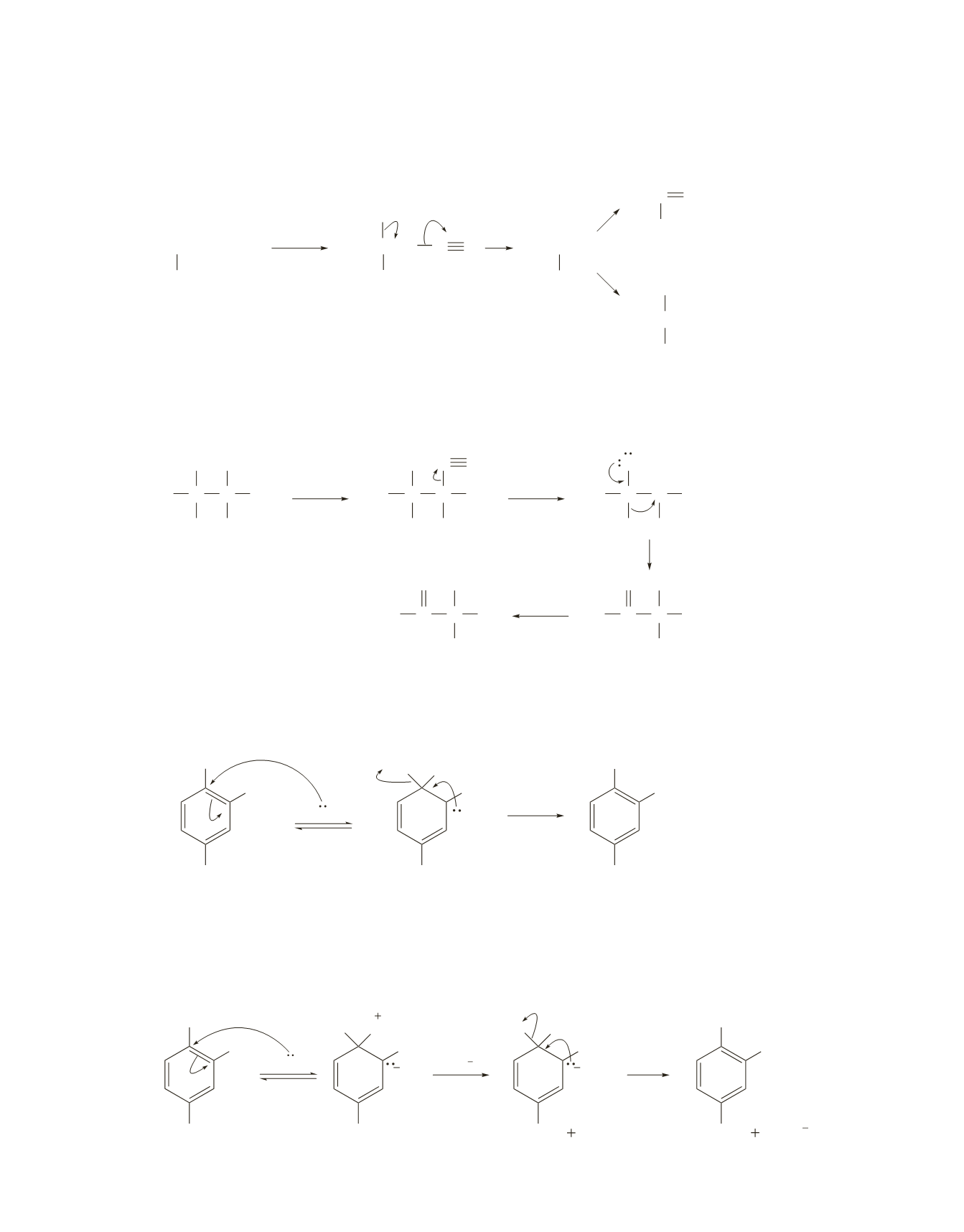
644 Chapter 18
Copyright © 2017 Pearson Education, Inc.
98.
a.
The alkyl diazonium ion is very unstable. Loss of N
2
and a 1,2-hydride shift forms a
tert
-butyl carbo-
cation, which can undergo either substitution or elimination.
CH
3
CHCH
2
NH
2
CH
3
NaNO
2
HCl
+
CH
3
CCH
3
CH
3
CH
3
CCH
2
N N
H
CH
3
+
+
CH
3
C CH
2
CH
3
H
3
O
+
+
H
3
O
+
CH
3
CCH
3
CH
3
OH
H
2
O
H
2
O
b.
The cation formed from the diazonium ion will undergo a pinacol-like rearrangement. (See Chapter 10,
Problem 92.)
CH
3
C C CH
3
OH NH
2
CH
3
NaNO
2
HCl
CH
3
C C CH
3
OH
CH
3
N
CH
3
N
+
CH
3
C C CH
3
OH
CH
3
CH
3
+
+
N
2
CH
3
C C CH
3
OH CH
3
CH
3
+
CH
3
C C CH
3
O CH
3
CH
3
+
H
+
CH
3
99.
A chloro group is a better leaving group than the ammonium group, so the product is formed without
hydroxide ion catalysis.
Cl
NO
2
NO
2
NO
2
NHR
2
Cl
NO
2
+
NHR
2
+
−
NO
2
NO
2
+
Cl
−
R
2
NH
A methoxy group is a poorer leaving group than the ammonium group, so the ammonium group is elimi-
nated, reforming starting materials. If hydroxide ion is added to the solution, hydroxide ion converts
the ammonium group into an amino group. Because the amino group is a poorer leaving group than the
methoxy group, the methoxy group will be eliminated.
NO
2
CH
3
O
NO
2
NO
2
NO
2
NO
2
NO
2
NO
2
NR
2
NO
2
NHR
2
CH
3
O
NR
2
CH
3
O
HO
H
2
O
CH
3
O
R
2
NH
















