
Chapter 18 633
Copyright © 2017 Pearson Education, Inc.
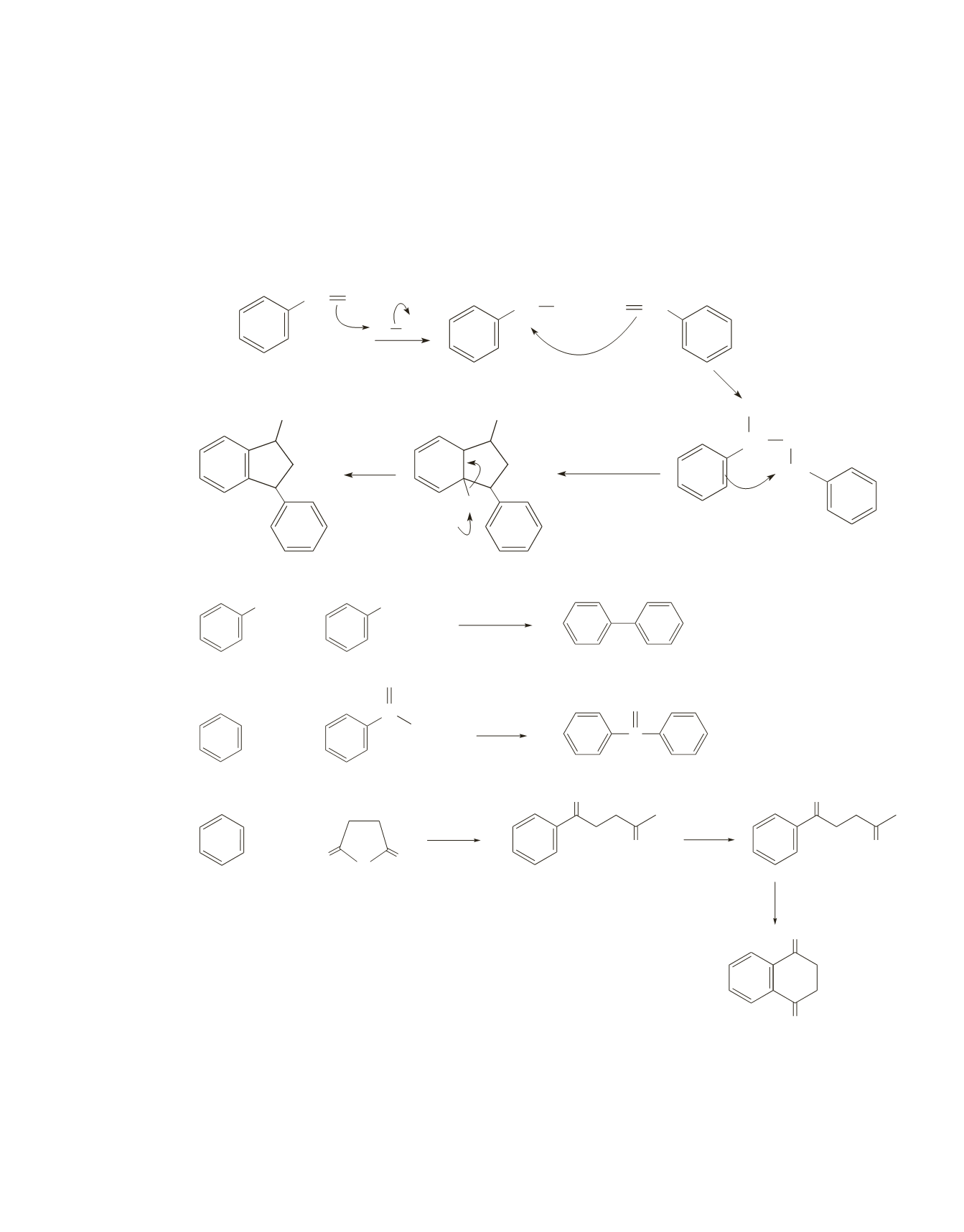
b.
As in part
a
, an electrophile is formed that can react in either an intermolecular reaction or an intra-
molecular reaction. Seeing that the product of the reaction has two benzene rings and that there are
twice as many carbons in the product as in the reactant indicates that two reactant molecules react in an
intermolecular reaction. In this case, the intermolecular reaction is favored, because the intramolecular
reaction would lead to a highly strained three-membered ring. The electrophile that is formed in the
intermolecular reaction can add to the benzene ring in an intramolecular reaction to form a stable five-
membered ring.
CH CH
2
CH CH
3
an intermolecular reaction
an intramolecular
reaction
H Cl
+
CH CH
2
CH
3
CH
+
CH CH
2
CH
3
H
+
..
B
CH
3
80.
a.
+
B(OR)
2
Br
PdL
2
HO
−
+
1. AlCl
3
Cl
C
O
C
O
2. H
2
O
c.
3
1. AlCl
3
1. AlCl
2
2. H O
O O
O
O
O
O
+
O
Cl
O
SOCl
2
2
2. H O
O
O
−
b.
















