
632 Chapter 18
Copyright © 2017 Pearson Education, Inc.
b.
O
NH
2
NH
2
HO
−
,
1. HSCH
2
CH
2
CH
2
SH/HCl
2. H
2
, Raney Ni
The reason there are three ways to carry out the reaction in part
a
but only two ways to carry it out in part
b
is
because only a carbonyl group adjacent to a benzene ring can be reduced to a methylene group by H
2
, Pd
>
C.
79.
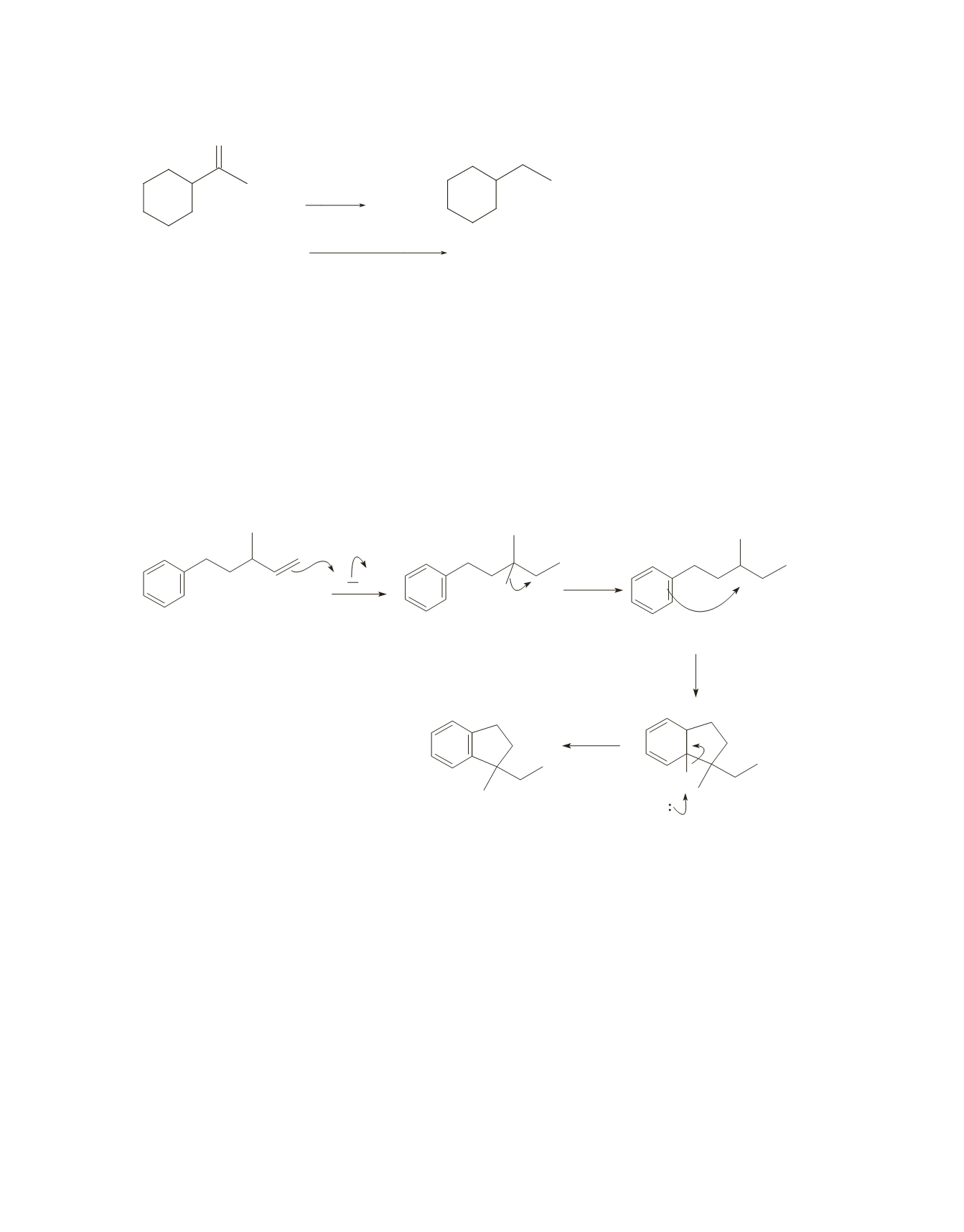
a.
HCl adds to the alkene, forming a secondary carbocation that undergoes a 1,2-hydride shift to form a
tertiary carbocation. The tertiary carbocation is an electrophile that can add either to the double bond
in a second molecule of the reactant (in an intermolecular reaction) or to the benzene ring in the same
molecule of the reactant (in an intramolecular reaction).
The intramolecular reaction is favored because it forms a stable five-membered ring. (See Section 9.16
in the text.) After the electrophile adds to the benzene ring, a base (B:) in the reaction mixture removes
a proton and the aromaticity of the benzene ring is restored.
H
+
B
H Cl
H
HB
+
+
+
+
an intramolecular reaction
















