
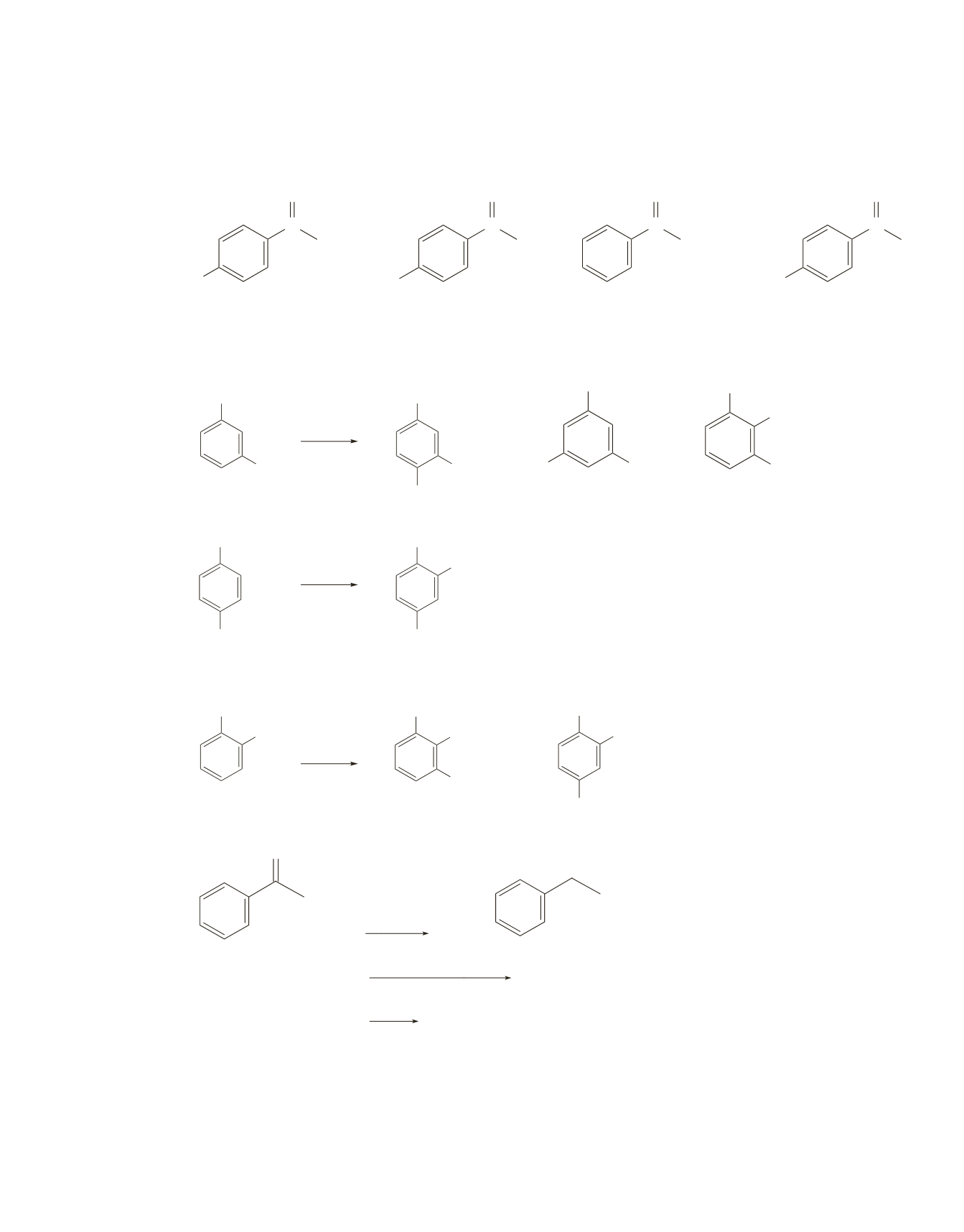
Chapter 18 631
Copyright © 2017 Pearson Education, Inc.
76.
The greater the electron-withdrawing ability of the para substituent, the greater the
K
eq
for hydrate forma-
tion. (See Section 16.9 in the text.)
O
2
N
C
O
CH
3
>
Cl
C
O
CH
3
>
C
O
CH
3
CH
3
O
C
O
CH
3
>
77.
The original compound was 1,3-dibromobenzene. 1,4-Dibromobenzene would form only one product, and
1,2-dibromobenzene would form only two.
Br
Br
HNO
3
Br
Br
NO
2
Br
HNO
3
Br
Br
Br
Br
NO
2
Br
NO
2
H
2
SO
4
H
2
SO
4
HNO
3
Br
Br
1,3-dibromobenzene
1,4-dibromobenzene
1,2-dibromobenzene
Br
Br
NO
2
Br
Br
O
2
N
Br
Br
NO
2
H
2
SO
4
+
+
+
minor product
78.
a.
O
H
2
Pd/C
NH
2
NH
2
HO
−
,
2. H
2
, Raney Ni
1. HSCH
2
CH
2
CH
2
SH/HCl
















