
626 Chapter 18
Copyright © 2017 Pearson Education, Inc.
63.
The rate-determining step in the S
N
1 reaction is formation of the tertiary carbocation. An electron-donating
substituent stabilize the carbocation and causes it to be more easily formed. An electron-withdrawing
substituent will destabilizes the carbocation and causes it to be less easily formed.
CH
3
CCH
3
Br
OCH
2
CH
3
>
CH
3
CCH
3
Br
OCCH
3
>
O
CH
3
CCH
3
Br
CH
2
CH
2
CH
3
>
CH
3
CCH
3
Br
CHClCH
3
>
CH
3
CCH
3
Br
SO
3
H
64.
The signal at 7 ppm that integrates to 5H indicates a monosubstituted benzene ring. A monosubstituted
benzene ring contains six carbons and five hydrogens. Subtracting these atoms from the molecular formula
of C
13
H
20
gives us a substituent with 7 carbons and 15 hydrogens. The compound shown below has the
correct number of carbons and hydrogens that will give two singlets, with one (9H) having 1.5 times the
area of the second (6H).
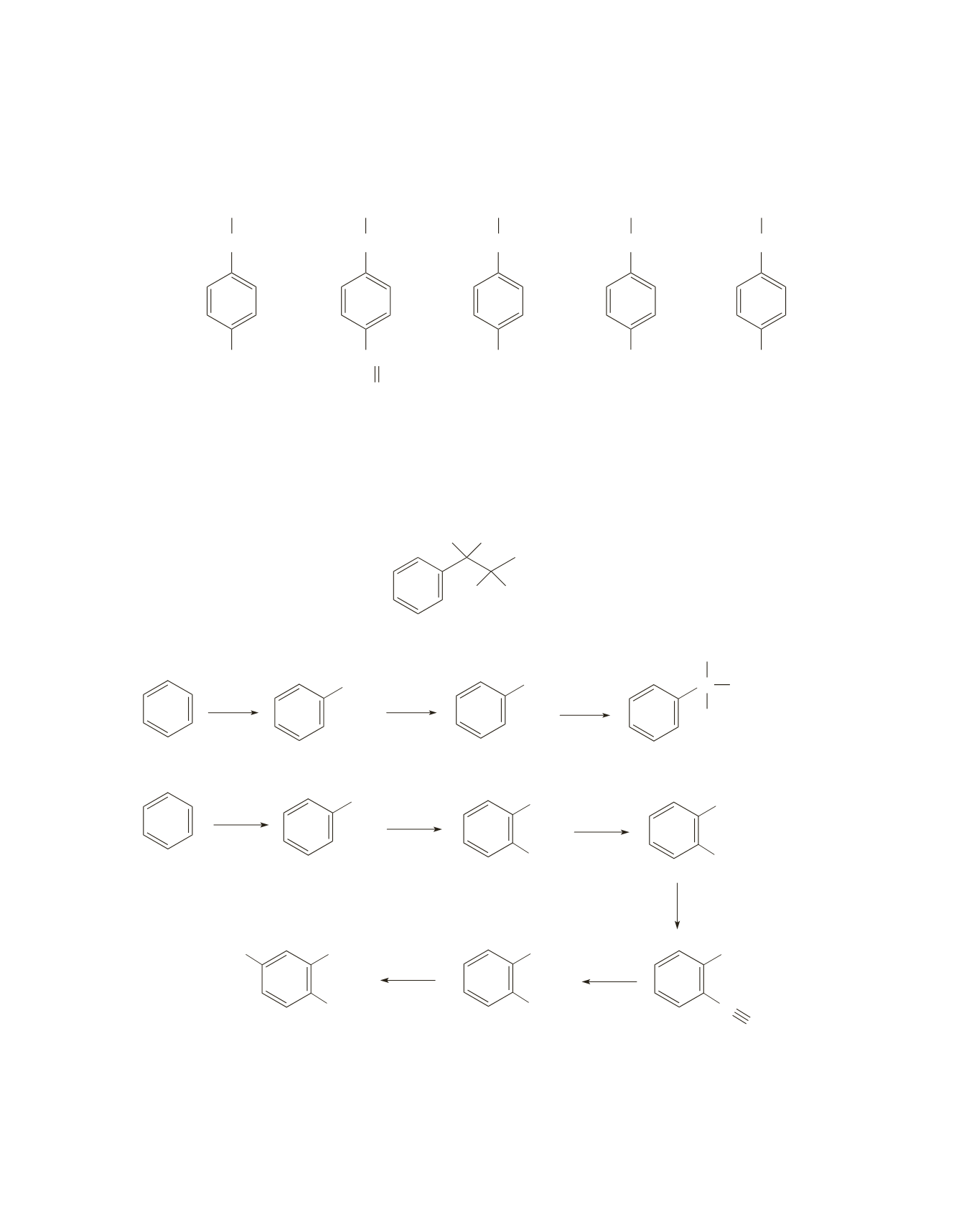
65.
a.
NO
2
NH
2
N
CH
3
CH
3
CH
3
+
I
–
HNO
3
H
2
SO
4
H
2
, Pd/C
CH
3
I
excess
K
2
CO
3
see page 405
of the text
b.
CH
3
OH
O
2
N
CH
3
OH
CH
3
N
N
CH
3
NH
2
CH
3
NO
2
CH
3
+
Cl
−
AlCl
3
CH
3
Cl
HNO
3
H
2
SO
4
H
2
,
Pd
/
C
NaNO
2
HCl
0 °
C
Cu
2
O
Cu
(
NO
3
)
2
,
H
2
O
or
H
3
O
+
,
HNO
3
H
2
SO
4
















