
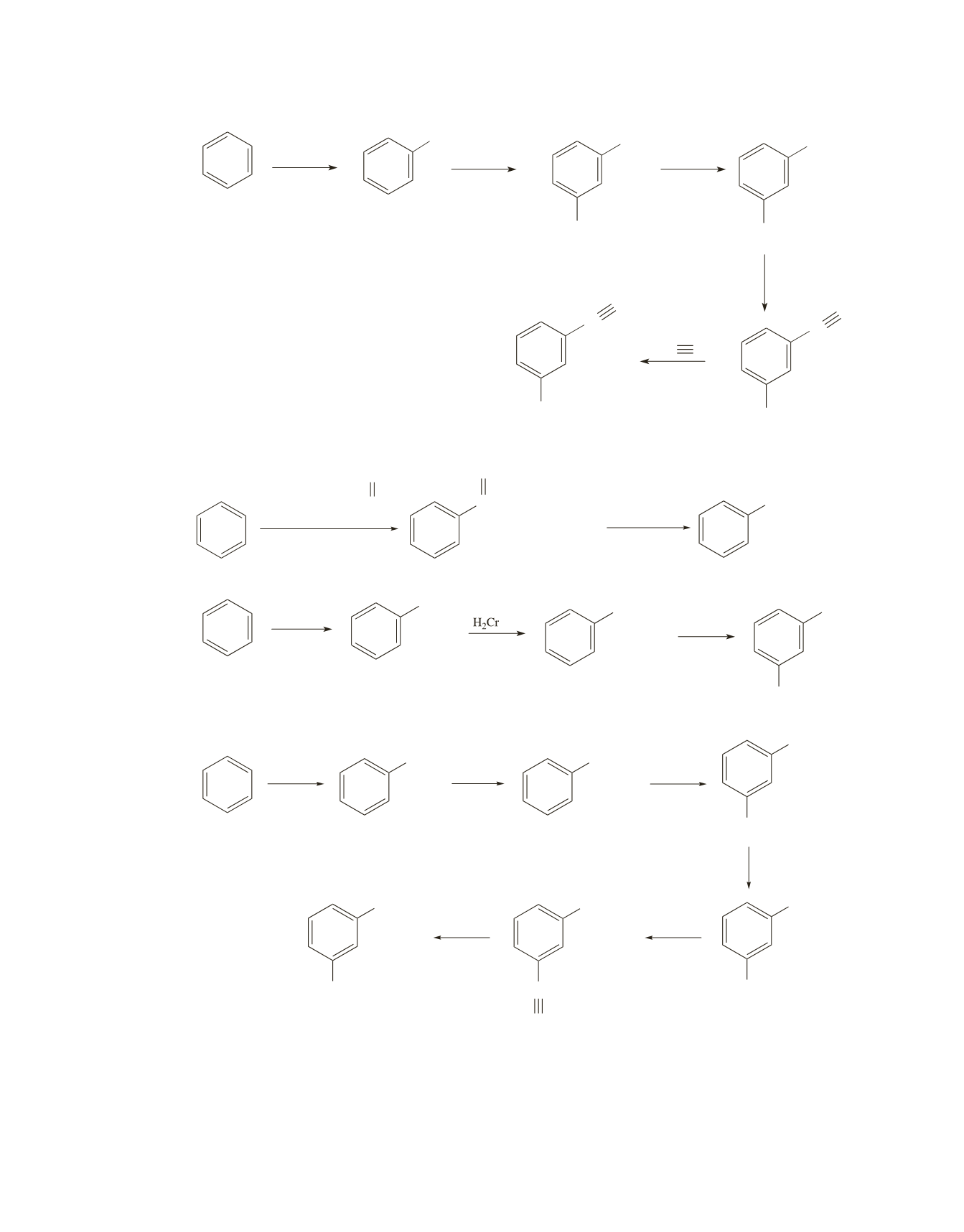
Chapter 18 621
Copyright © 2017 Pearson Education, Inc.
c.
+
Cl
−
NO
2
NO
2
Br
NH
2
Br
N
Br
N
C
Br
N
HNO
3
H
2
SO
4
Br
2
FeBr
3
0
°
C NaNO
2
HCl
CuC N
H
2
, Pd/C
d.
1. CH
3
CH
2
CH
2
CH
2
CCl
O
AlCl
3
2. H
2
O
CCH
2
CH
2
CH
2
CH
3
O
or
H
2
, Pd/C
CH
2
CH
2
CH
2
CH
2
CH
3
H
2
NNH
2
HO
−
,
e.
CH
3
COOH
COOH
Br
CH
3
Cl
AlCl
3
FeBr
3
Br
2
O
4
f.
CH
3
Cl
CH
3
COOH
COOH
NO
2
H
2
SO
4
COOH
NH
2
COOH
N
COOH
OH
N
NaNO
2
HCl
0
°
C
AlCl
3
HNO
3
H
2
CrO
4
+
H
2
, Pd/C
Cu
2
O
Cu
(
NO
3
)
2
,
H
2
O
or
H
3
O
+
,
















