
Chapter 18 609
Copyright © 2017 Pearson Education, Inc.
26.
Yes, the advice is sound.
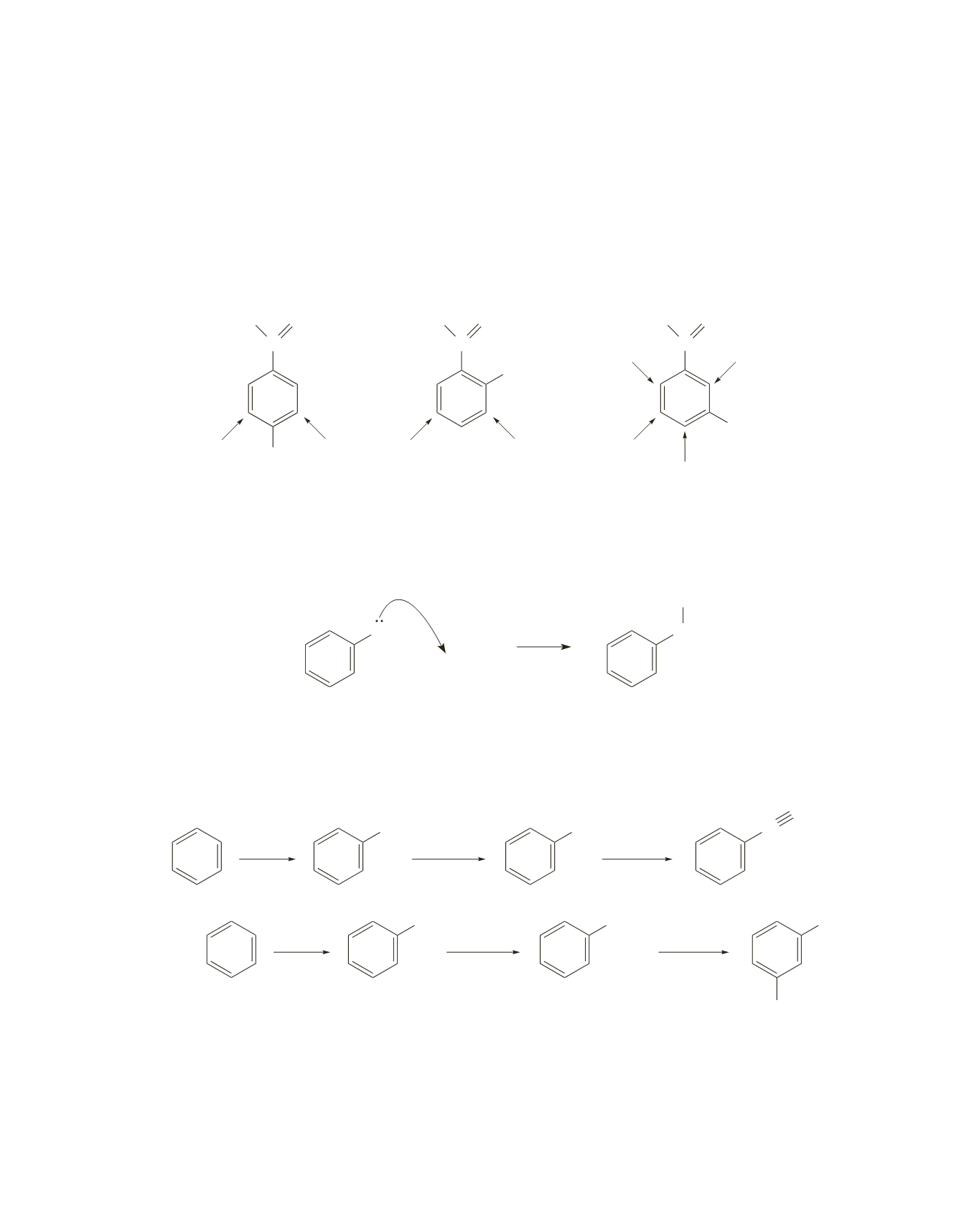
The para isomer will form one product, because the formyl and ethyl groups both direct to the same
positions and both positions result in the same product.
The ortho isomer will form two products, because the formyl and ethyl groups both direct to the same
positions but different products are obtained from each position.
The meta isomer will form four products, because the formyl and ethyl groups direct to four different
positions and a different product is obtained from each position.
CH
2
CH
3
CH
2
CH
3
CH
2
CH
3
low yield
C
O H
C
O H
C
O H
27.
Solved in the text.
28.
FeBr
3
will complex with the amino group, converting it into a meta director. The NH
2
group is a strongly
activating substituent, so a Lewis acid is not needed.
_
+
NH
2
FeBr
3
FeBr
3
+
NH
2
29.
Because a diazonium ion is electron withdrawing, it deactivates the benzene ring toward electrophilic
aromatic substitution. A deactivated benzene ring is too unreactive to undergo an electrophilic substitution
reaction at the cold temperature necessary to keep the benzenediazonium ion from decomposing.
30.
NH
2
NO
2
N
N
+
Cl
−
HNO
3
H
2
SO
4
NaNO
2
HCl, 0
°
C
H
2
, Pd/C
31.
a.
COOH
COOH
CH
3
CH Cl
3
AlCl
3
HNO
3
H
2
CrO
4
H
2
SO
4
NO
2
















