
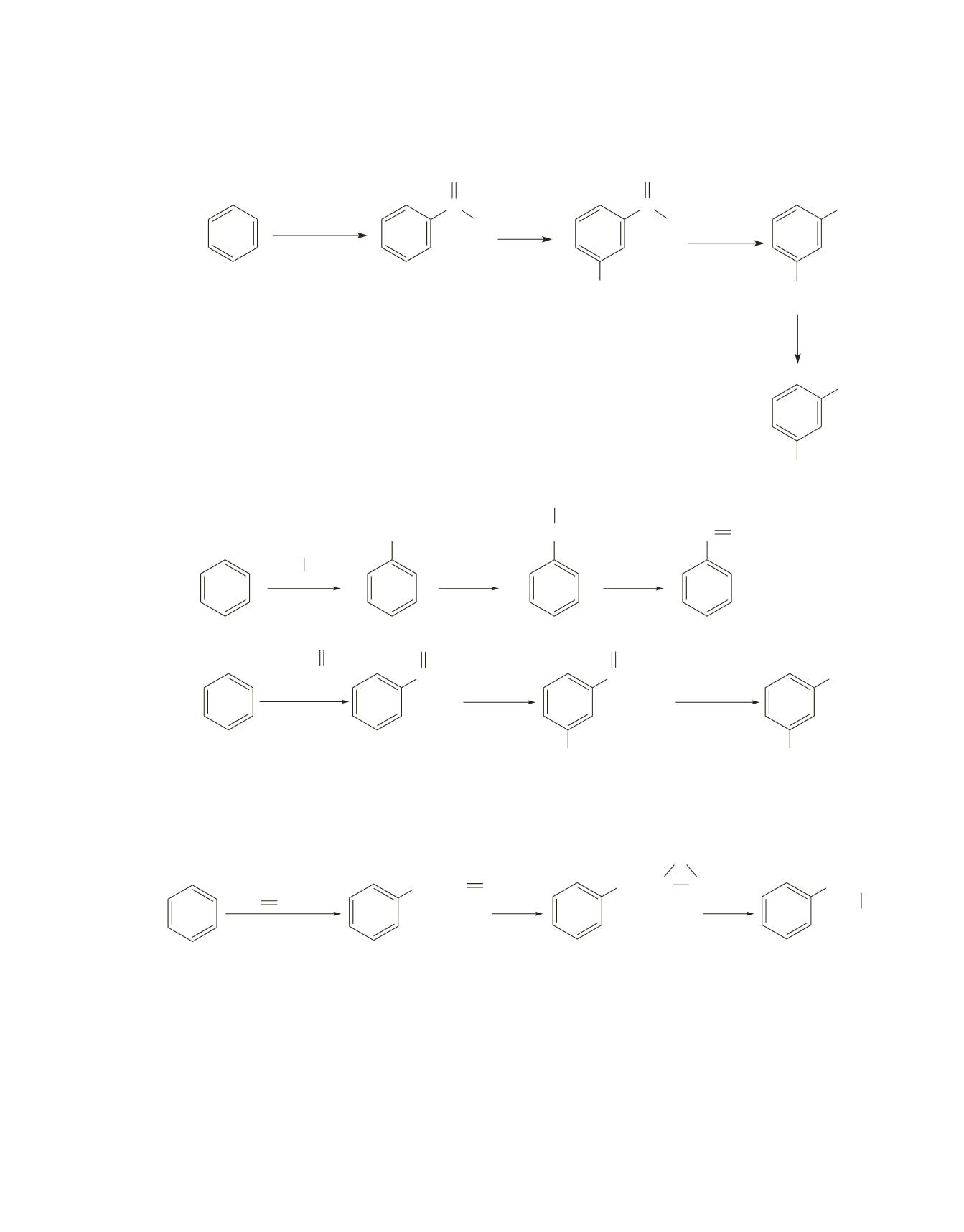
Chapter 18 607
Copyright © 2017 Pearson Education, Inc.
f.
Note that benzaldehyde is formed by the Gatterman–Koch reaction (page 877 in the text).
C
H
O
Br
2
FeBr
3
CH
3
B(OR)
2
PdL
2
, NaOH
CO, HCl
high pressure
AlCl
3
, CuCl
C
O
Br
CH
3
Br
CH
3
CH
3
or
(CH
3
)
2
CuLi
H
H
2
NNH
2
HO
−
,
or
H
2
, Pd/C
g.
CH
3
CHCH
3
NBS,
peroxide
CH
3
CCH
3
Br
CH
3
CHCl
AlCl
3
CH
3
tert
-BuO
−
CH
3
C CH
2
h.
CCH
2
CH
3
O
CH
3
CH
2
CCl
AlCl
3
O
2. H
2
O
1.
CH
2
CH
2
CH
3
FeBr
3
Br
2
CH
2
CH
2
CH
3
Br
or
H
2
, Pd/C
H
2
NNH
2
HO
−
,
CCH
2
CH
3
O
CCH
2
CH
3
O
Br
CH
2
CH
2
CH
3
Br
CH
3
CH
2
CCl
AlCl
3
O
2. H
2
O
1.
Br
2
FeBr
3
or
H
2
, Pd/C
H
2
NNH
2
HO
−
,
h.
a.
i.
Notice that in the second step of the synthesis, epoxidation followed by the addition of hydride ion
(page 483) is used to add water to the double bond in order to avoid the carbocation rearrangement that
would occur with the acid-catalyzed addition of water.
CH
2
CHCH
2
Cl
AlCl
3
CH
2
CH CH
2
CH
2
CHCH
3
OH
1. NaH
2. HCl
MCPBA
CH
2
CH
O
CH
2
















