
Chapter 18 605
Copyright © 2017 Pearson Education, Inc.
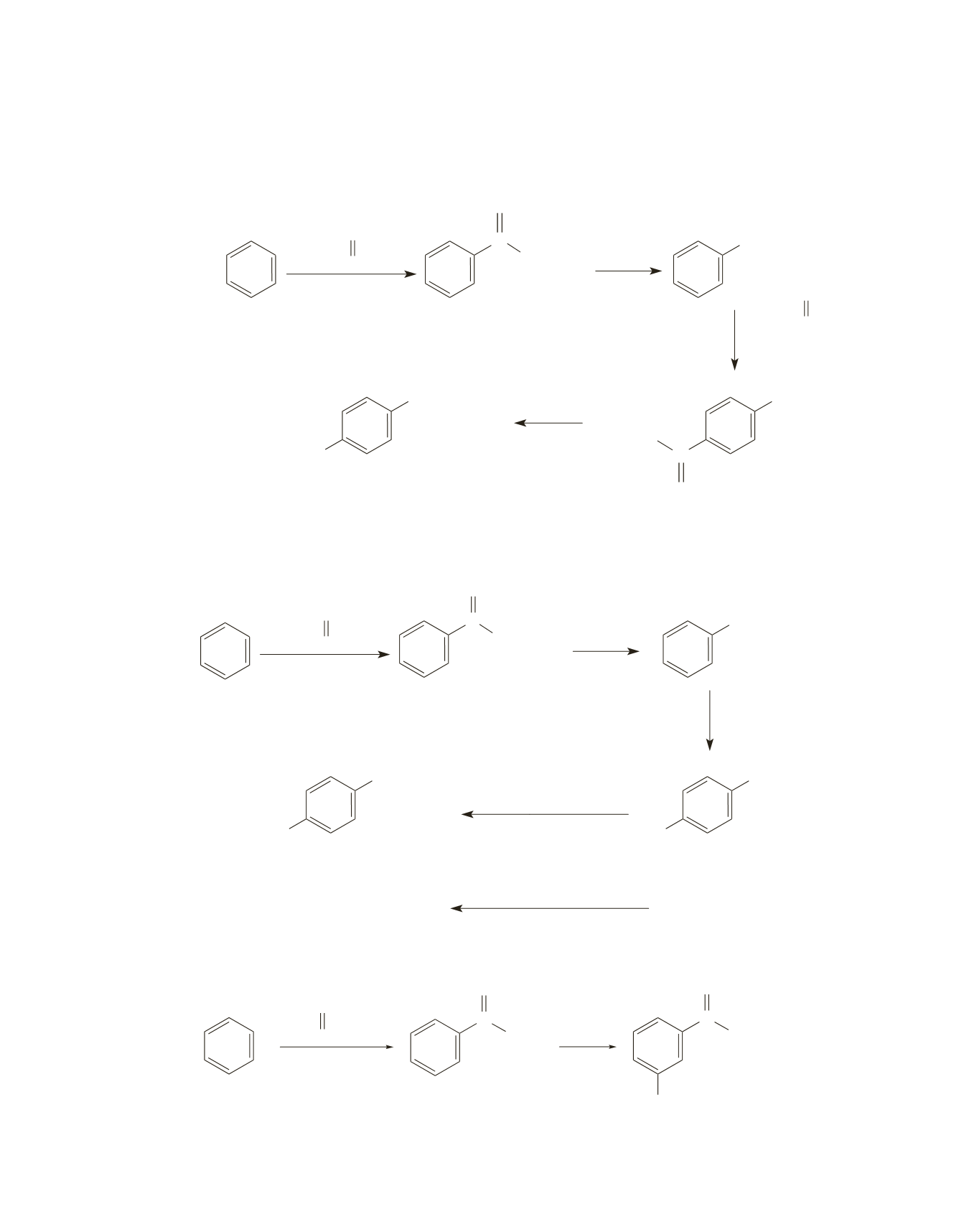
22.
a.
Unlike
m
-dipropylbenzene that requires a coupling reaction to be used in its synthesis, a coupling
reaction does not have to be used in the synthesis of
p
-dipropylbenzene. The propyl group is an ortho/
para director, so propylbenzene can undergo a Friedel–Crafts reaction.
C
CH
2
CH
3
O
1.
2.
CH
3
CH
2
CCl/AlCl
3
O
H
2
O
H
2
Pd/C
CH
2
CH
2
CH
3
CH1.
3
CH
2
CCl/AlCl
3
O
2. H
2
O
CH
2
CH
2
CH
3
C
CH
3
CH
2
O
H
2
Pd/C
CH
2
CH
2
CH
3
CH
3
CH
2
CH
2
b.
A coupling reaction can be used in the synthesis of
p
-dipropylbenzene. Notice that bromination occurs
after the reduction of the carbonyl group. In contrast, bromination occurs before the reduction reaction
in the synthesis of
m
-dipropylbenzene.
PdL
2
, NaOH
CH
3
CH
2
CH
2
B(OR)
2
FeBr
3
Br
2
CH
2
CH
2
CH
3
CH
3
CH
2
CH
2
CH
2
CH
2
CH
3
Br
CH
2
CH
2
CH
3
C
O
(CH
3
CH
2
CH
2
)
2
CuLi
or
1.
2.
CH
3
CH
2
CCl/AlCl
3
O
H
2
O
H
2
Pd/C
CH
2
CH
3
23.
Notice that in all three syntheses, the Friedel–Crafts reaction has to be done first. Both NO
2
and SO
3
H are
meta directors, and a Friedel–Crafts reaction cannot be carried out if a meta director is on the ring.
a.
2. H
2
O
2. H
2
O
HNO
3
H
2
SO
4
H
2
SO
4
1. CH
3
3
CCl/AlCl
O
1. CH
3
3
CH
2
CCl/AlCl
O
CH
2
CH
3
C
O
CH
3
C
O
CH
2
CH
3
C
O
CH
3
C
O
NO
2
















