
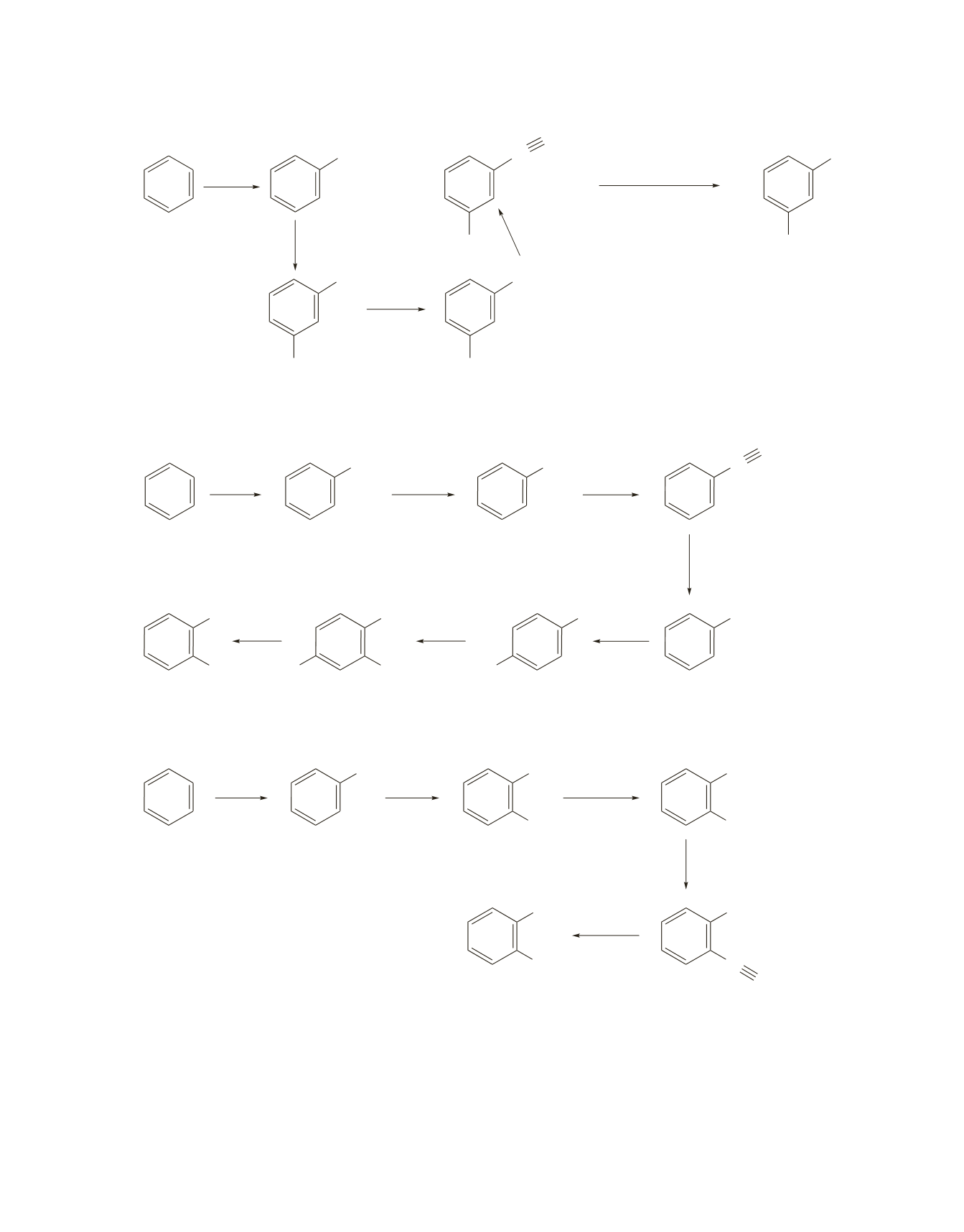
610 Chapter 18
Copyright © 2017 Pearson Education, Inc.
b.
OH
Br
H
3
O
+
H
2
or
Cu(NO
3
)
2
, H
,
2
O
Cu
2
O
Br
−
+
N
N
Br
NO
Br
HNO
3
H
2
SO
4
2
FeBr
3 2
Br
NO
2
NH
Br
2
Pd/C
NaNO
2
HCl, 0
°
C
c.
The first approach is longer, but it will generate a higher yield of the ortho isomer.
or
Cl
−
+
N
N
NO
2
NH
2
OH
OH
HO
3
S
OH
HO
3
S
Cl
OH
Cl
Cu(NO
3
)
2
,
H
2
O
HNO
3
H
2
SO
4
NaNO
2
HCl, 0
°
C
Cu
2
O
H
2
SO
4
Cl
2
FeCl
3
H
3
O
+
or
H
3
O
+
,
H
2 ,
Pd/C
+
Cl
Cl
NO
2
Cl
N
N
Cl
OH
Cl
NH
2
Cl
−
Cl
2
FeCl
3
HNO
3
H
2
SO
4
NaNO
2
HCl
0
°
C
Cu
2
O
Cu(NO
3
)
2
,
H
2
O
or
H
3
O
+
,
H
2
, Pd/C
d.
The nitro group cannot be placed on the benzene ring first, because a Friedel–Crafts reaction cannot
be carried out on a ring with a meta director. Because formyl chloride is too unstable to be purchased,
benzaldehyde is prepared by the Gatterman–Koch reaction (page 884 in the text).
















