
Chapter 18 615
Copyright © 2017 Pearson Education, Inc.
41.
a.
BrCH
2
CH
2
CH
2
CHCH
2
CH
3
O CH
2
CH
3
NaH
OH
CH
2
CH
2
CH
2
CHCH
3
Cl
CH
3
AlCl
3
b.
BrCH
2
CH
2
CH
2
CHCH
2
CH
3
O CH
2
CH
3
NaH
OH
CH
2
CH
2
CH
2
CHCH
3
Cl
CH
3
AlCl
3
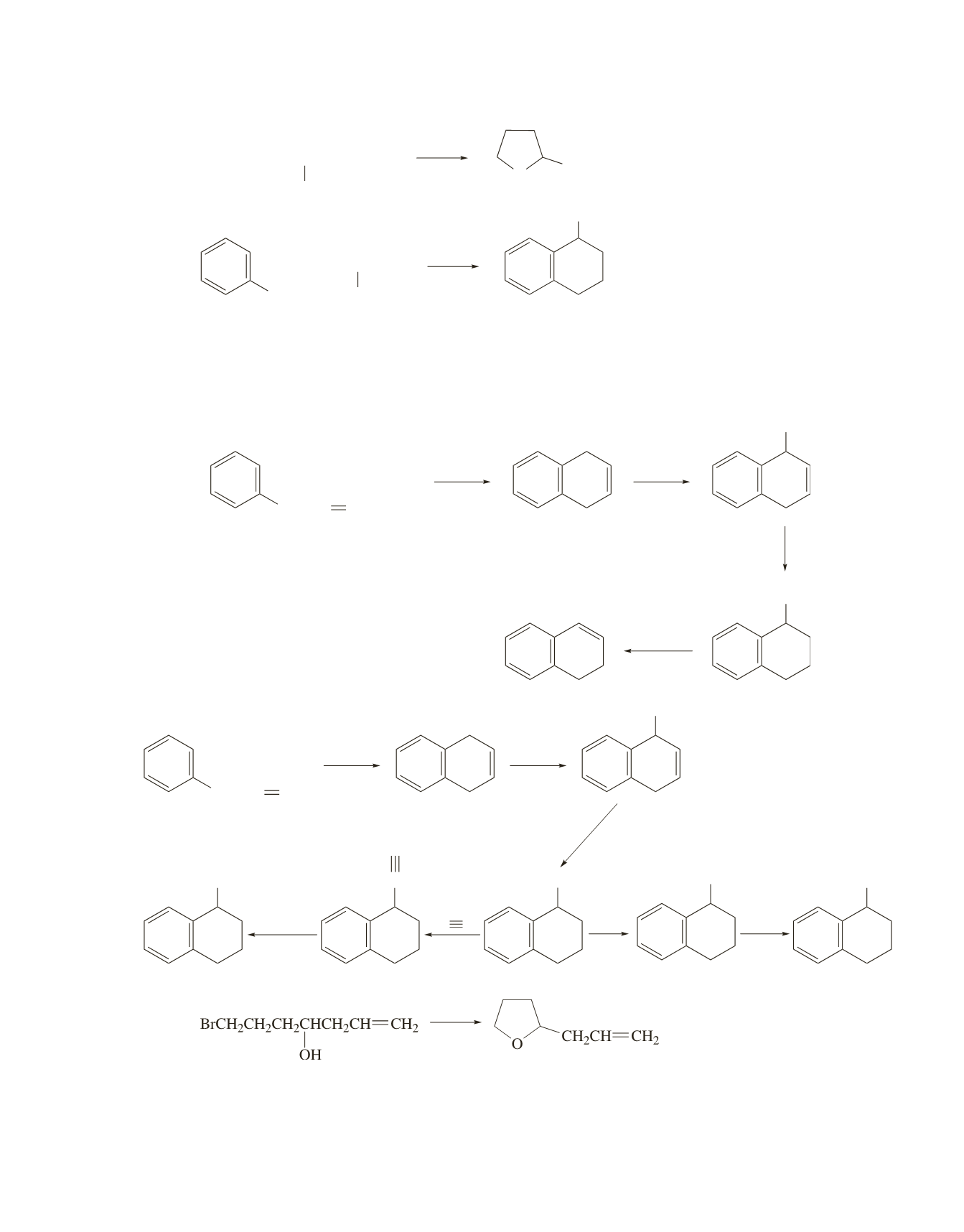
c.
In this synthesis of
c
and
d
, a primary alkyl halide cannot be used in the first step because the
carbocation will rearrange to a secondary carbocation, which will result in the formation of a methyl-
substituted five-membered ring. A primary allyl halide can be used because the initially formed allyl
cation is stabilized by electron delocalization.
CH
2
CH CHCH
2
Cl
NBS,
peroxide
Br
H
2
, Pd/C
Br
tert
-BuO
AlCl
3
−
d.
CH
2
CH CHCH
2
Cl
NBS,
peroxide
Br
Mg
MgBr
H
2
, Pd/C
Br
AlCl
3
C N
−
C
N
H
2
O, HC
Et
2
O
1. CO
2
2. HCl
l
COOH
COOH
e.
NaH
















