
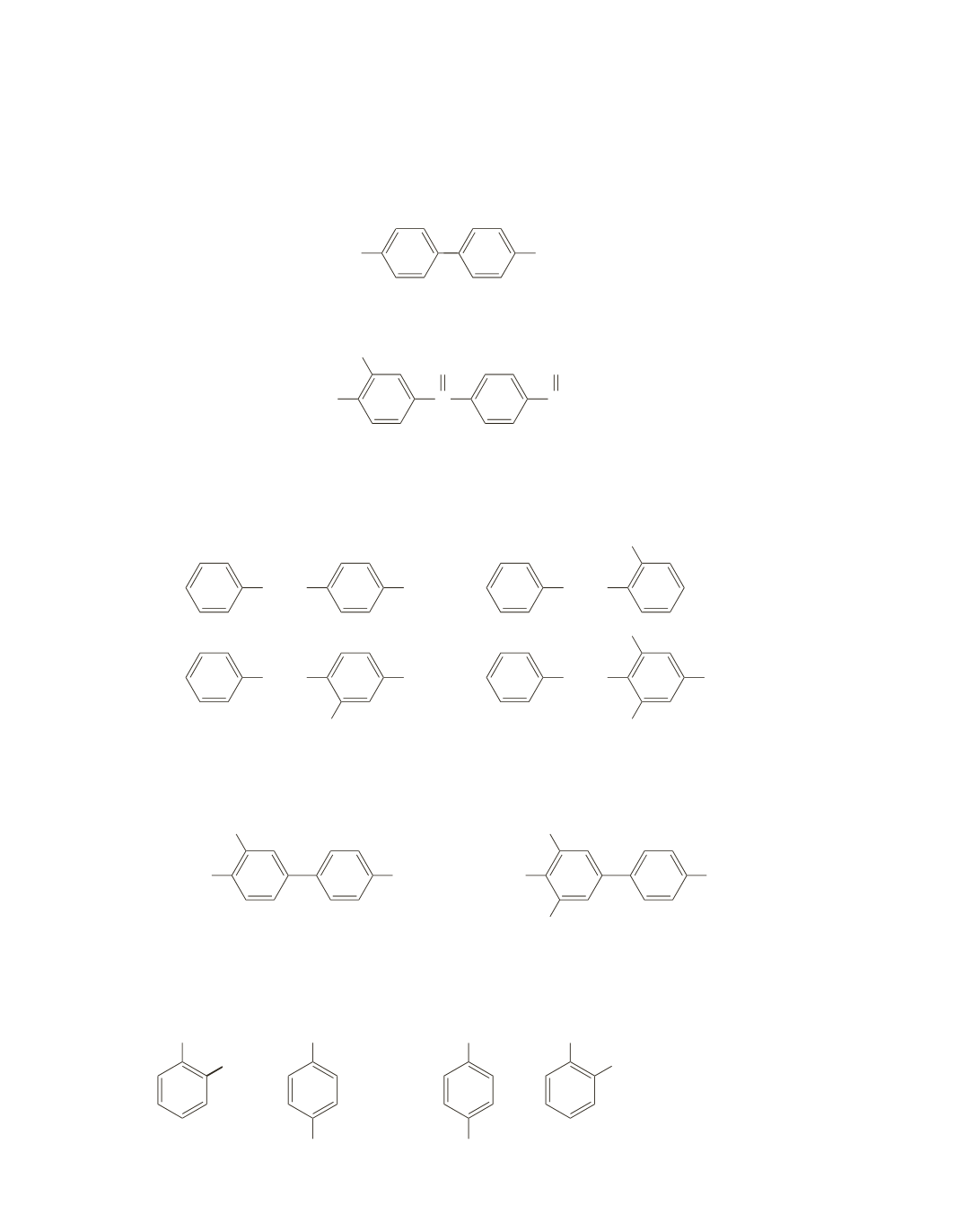
604 Chapter 18
Copyright © 2017 Pearson Education, Inc.
20.
For each compound, determine which benzene ring is more highly activated. The more highly activated
ring is the one that undergoes electrophilic aromatic substitution.
a.
Because the left-hand ring is weakly activated and the right-hand ring is strongly activated, electro-
philic aromatic substitution occurs on the right-hand ring.
CH
3
OCH
3
b.
Because the left-hand ring is activated and the right-hand ring is deactivated, electrophilic aromatic
substitution occurs on the left-hand ring.
Br
CH
3
C
O
COCH
3
O
c.
Because the right-hand ring is highly activated and a catalyst is employed, monobrominated, dibromi-
nated, and tribrominated compounds can be obtained. However, because only one equivalent of Br
2
is
available, there is not enough bromine to brominate all the reactive positions. Therefore, a mixture of
unreacted starting material and the four products shown below will be obtained.
CH
2
O
Br
CH
2
O
Br
CH
2
O
Br
CH
2
O
Br
Br
Br
Br
d.
Because the left-hand ring is highly activated and a catalyst is employed, both ortho positions can be
brominated. Because not enough Br
2
is available to brominate both reactive positions, a mixture of
unreacted starting material and the two products shown below will be obtained.
NO
2
Br
Br
CH
3
O
NO
2
Br
CH
3
O
21.
No reaction will occur in
a
and
c
, because a Friedel–Crafts reaction cannot be carried out on a ring that
possesses a meta director.
a.
no reaction
c.
no reaction
OH
2
Br
OH
2
Br
b.
CH
3
CH
3
d.
+
+
CH
3
CH
3
















