
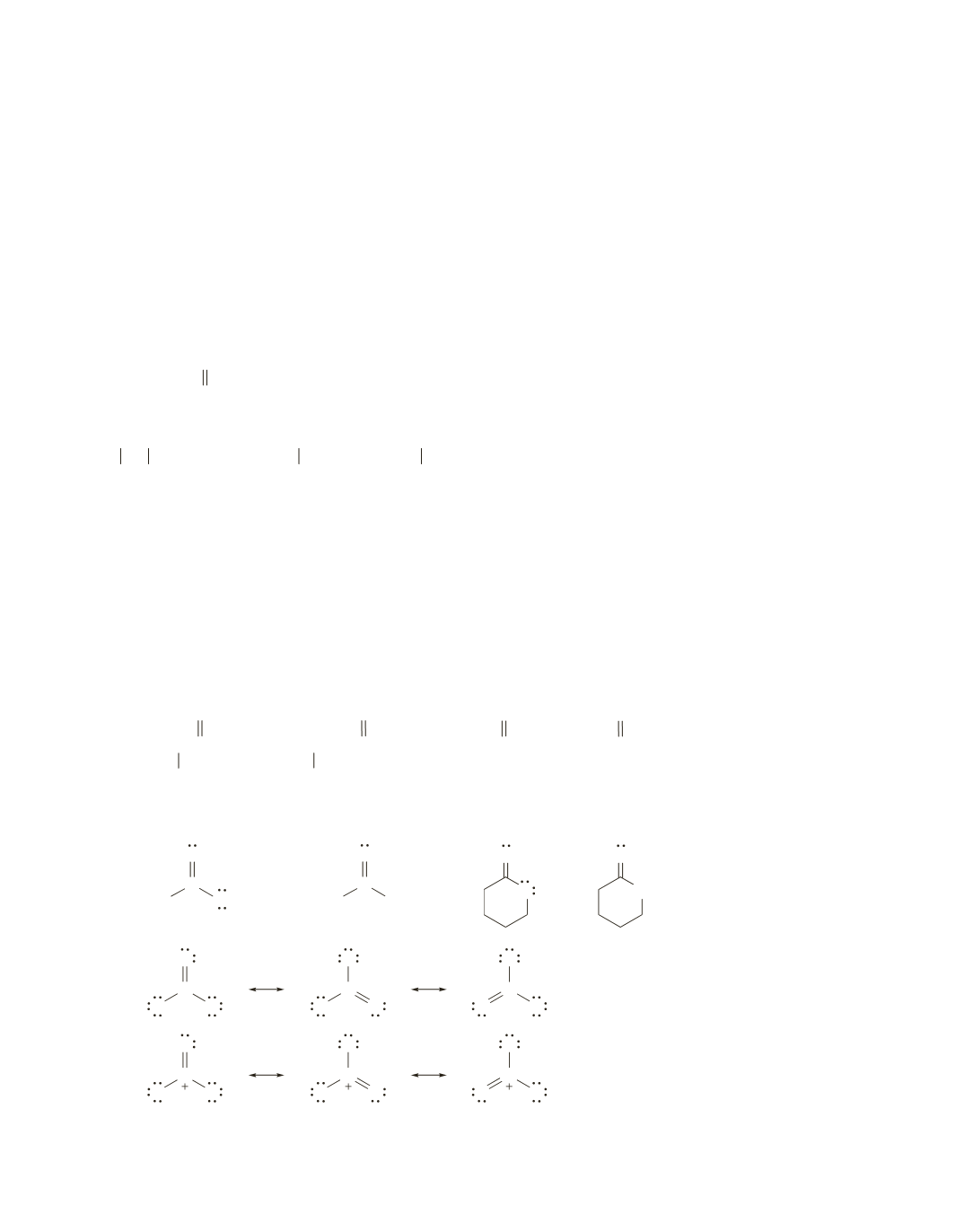
98
Chapter 2
Copyright © 2017 Pearson Education, Inc.
29.
Compare the acid strengths of the conjugate acids, recalling that a weaker acid has a stronger conjugate base.
a.
CH
3
O
-
because
CH
3
OH
is a weaker acid than
CH
3
SH.
b.
HO
-
because
H
2
O
is a weaker acid than
H
3
O
+
.
c.
NH
3
because
+
NH
4
is a weaker acid than
H
3
O
+
.
d.
CH
3
O
-
because
CH
3
OH
is a weaker acid than
CH
3
COOH.
30.
a.
CH
3
OCH
2
CH
2
OH
because its conjugate base has its negative charge stabilized by electron
withdrawal by the
CH
3
O
group.
b.
CH
3
CH
2
CF
2
CH
2
O
+
H
2
because oxygen is more electronegative than nitrogen.
c.
CH
3
CH
2
OCH
2
CH
2
OH
because the electron-withdrawing oxygen is closer to the OH group.
d.
CH
3
CH
2
COH
O
because the electron-withdrawing
C
“
O
is closer to the OH group.
31.
CH
2
CHCH
2
COOH >
F F
CH
2
CH
2
CH
2
COOH >
F
CH
3
CHCH
2
COOH >
F
CH
3
CH
2
CH
2
COOH
The first listed compound is the most acidic because it has two electron-withdrawing substituent that stabi-
lize the conjugate base.
The second listed compound is a stronger acid than the third listed compound because the fluorine in the
third compound is farther away from the
O
¬
H
bond, so the electron-withdrawing group will not be as
effective in stabilizing the conjugate base.
The compound on the far right does not have a substituent that withdraws electrons inductively, so it is the
least acidic of the four compounds.
32.
The weaker acid has a stronger conjugate base.
a.
CH
3
CHCO
−
O
Br
b.
CH
3
CHCH
2
CO
−
Cl
O
c.
CH
3
CH
2
CO
−
O
d.
CH
3
CCH
2
CH
2
O
−
O
33.
Solved in the text.
34.
a.
C
OH
CH
3
OCH
3
+
b.
C
OH
CH
3
NH
2
+
c.
OH
+
O
d.
OH
+
NCH
3
35.
a.
C
O
O O
−
−
C
O
O O
−
−
C
O
O O
−
−
b.
N
O
O O
−
−
N
O
O O
−
−
N
O
O O
−
−
















