
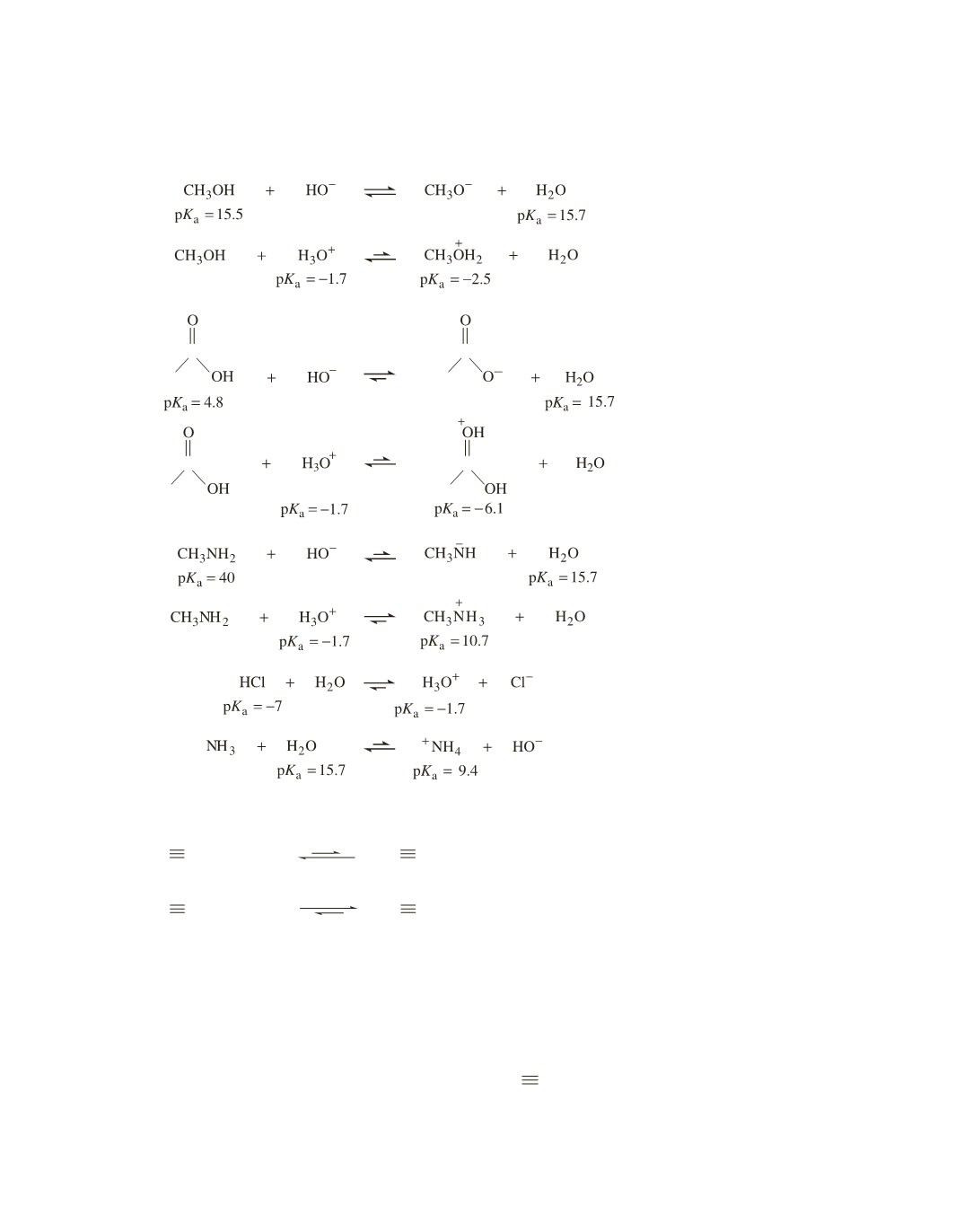
96
Chapter 2
Copyright © 2017 Pearson Education, Inc.
16.
Recall that the equilibrium favors reaction of the stronger acid to form the weaker acid. Because the
p
K
a
values in part
a
are similar, there will be similar amounts of reactants and products at equilibrium.
a.
C
CH
3
C
CH
3
C
CH
3
C
CH
3
b.
17.
Because a strong acid is more likely to lose a proton than a weak acid, the equilibrium favors loss of a
proton from the strong acid and formation of the weak acid.
a.
HC CH
+
HO
−
H
2
O
HC C
−
+
p
K
a
=
25
p
K
a
=
15.7
b.
HC CH
+
−
NH
2
HC C
−
+
p
K
a
=
25
NH
3
p
K
a
=
36
c.
-
NH
2
would be a better base because when it removes a proton, the equilibrium favors the products.
When
HO
-
removes a proton, the equilibrium favors the reactants.
18.
Each of the following bases will remove a proton from acetic acid in a reaction that favors products,
because each of these bases forms an acid that is a weaker acid than acetic acid.
HO CH
3
NH
2
−
−
HC C
The other three choices form an acid that is a stronger acid than acetic acid.
















