
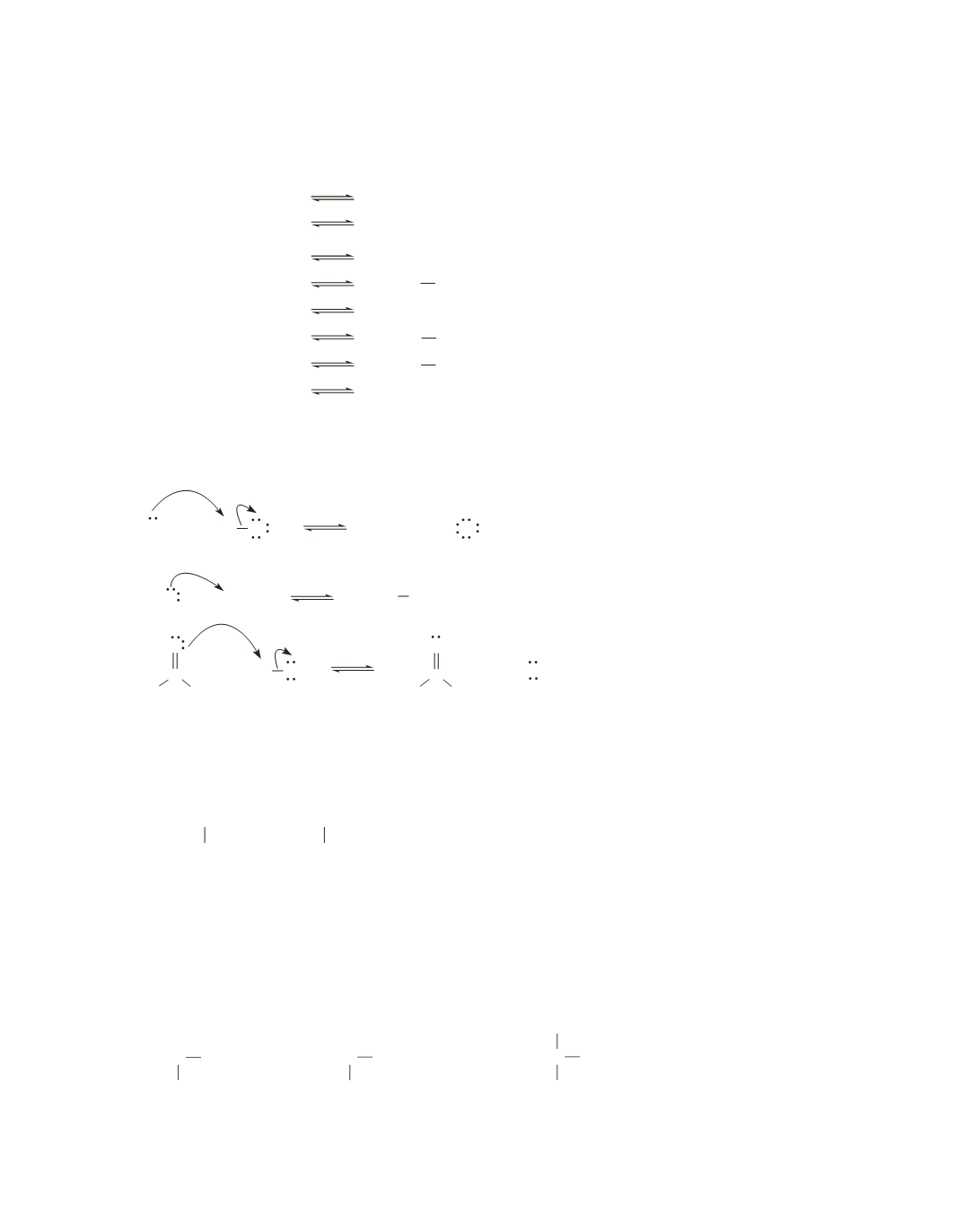
102 Chapter 2
Copyright © 2017 Pearson Education, Inc.
52.
a, b, c,
and
h
are Brønsted acids (proton-donating acids). Therefore, they react with
HO
-
by giving a
proton to it.
d, e, f,
and
g
are Lewis acids. They react with
HO
-
by accepting a pair of electrons from it.
a.
−
+
3
CH OH HO
−
+
3
2
CH O H O
b.
−
+
+
4
NH HO
+
3
2
NH H O
c.
−
+
+
3 3
CH NH HO
+
3 2
2
CH NH H O
d.
−
+
3
BF
HO
−
3
HO BF
e.
−
+
+
3
CH HO
3
CH OH
f.
−
+
3
FeBr
HO
−
3
HO FeBr
g.
−
+
3
AlCl
HO
−
3
HO AlCl
h.
−
+
3
CH COOH HO
−
+
3
2
CH COO H O
53.
The stronger base has the weaker conjugate acid.
a.
HO
-
b.
3
CH NH
−
c.
CH
3
O
-
d.
Cl
-
e.
−
CH
3
COO
f.
−
CH
3
CHBrCOO
54.
a.
+ NH
3
H Cl
NH
4
+ Cl
_
+
C
O
OH H
H
2
O + FeBr
3
+ H Cl
..
H
2
O FeBr
3
+
_
C
OH
OH H
+
+
..
Cl
. .
_
55.
a.
CCl
3
CH
2
OH
7
CHCl
2
CH
2
OH
7
CH
2
ClCH
2
OH
b.
The greater the number of electron-withdrawing chlorine atoms equidistant from the OH group, the
stronger the acid. (Notice that the larger the
K
a
,
the stronger the acid.)
56.
a.
CH
3
CH
2
CHCOOH >
Cl
CH
3
CHCH
2
COOH
Cl
> ClCH
2
CH
2
CH
2
COOH > CH
3
CH
2
CH
2
COOH
b.
An electron-withdrawing substituent makes the carboxylic acid more acidic, because it stabilizes its
conjugate base by decreasing the electron density around the oxygen atom. (Remember that the larger
the
K
a
,
the stronger the acid.)
c.
The closer the electron-withdrawing chloro substituent is to the acidic proton, the more it can decrease
the electron density around the oxygen atom because it has to exert its effect through fewer bonds.
Therefore, the closer it is, the more it stabilizes the conjugate base and increases the acidity of its con-
jugate acid.
57.
a.
+
CH
3
O BF
3
CH
3
..
_
b.
+
CH
3
O H
CH
3
..
_
+ Cl
c.
CH
3
N AlCl
3
H
H
+
_
58.
a.
HOCH
2
CH
2
CH
2
NH
3
+
b.
OCH
2
CH
2
CH
2
NH
2
−
b.
c.
















