
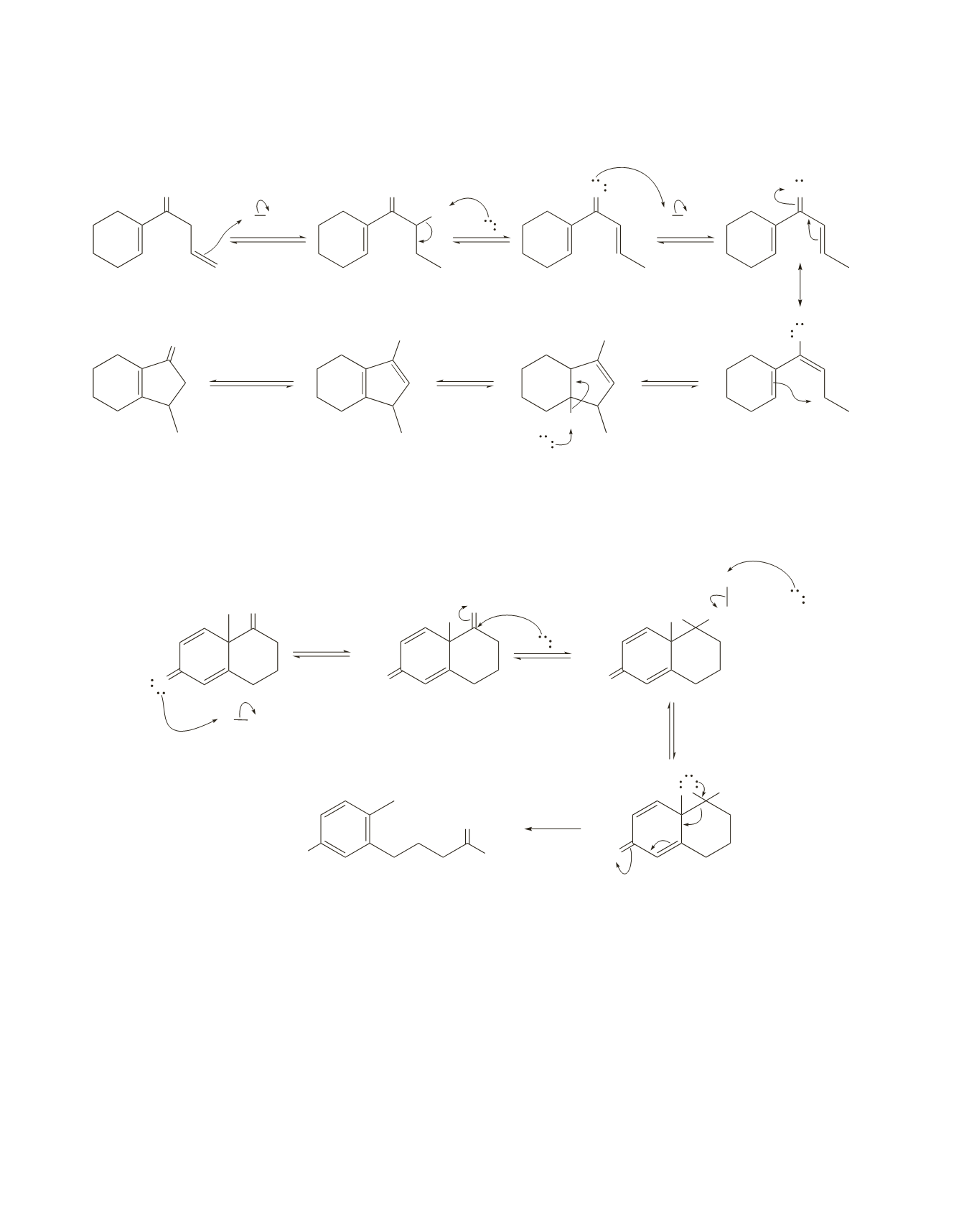
590 Chapter 17
Copyright © 2017 Pearson Education, Inc.
84.
a.
The mechanism starts with an isomerization that converts isolated double bonds to conjugated double
bonds.
O
O
O
OH
OH
OH
H
2
O
+
+
+
+
H
H
H OH
H
+
H OH
H
+
OH
H
3
O
+
H
2
O
O
tautomerization
b.
The carbonyl group on the left is protonated because electron delocalization causes it to be more basic
than the other carbonyl group.
O
O
H OH
H
+
HO
OH
O
+
H
3
O
+
H
2
O
HO
O
+
HO
+
OH
H
−
O
H
2
O
HO
+
OH
_
O
+
















