
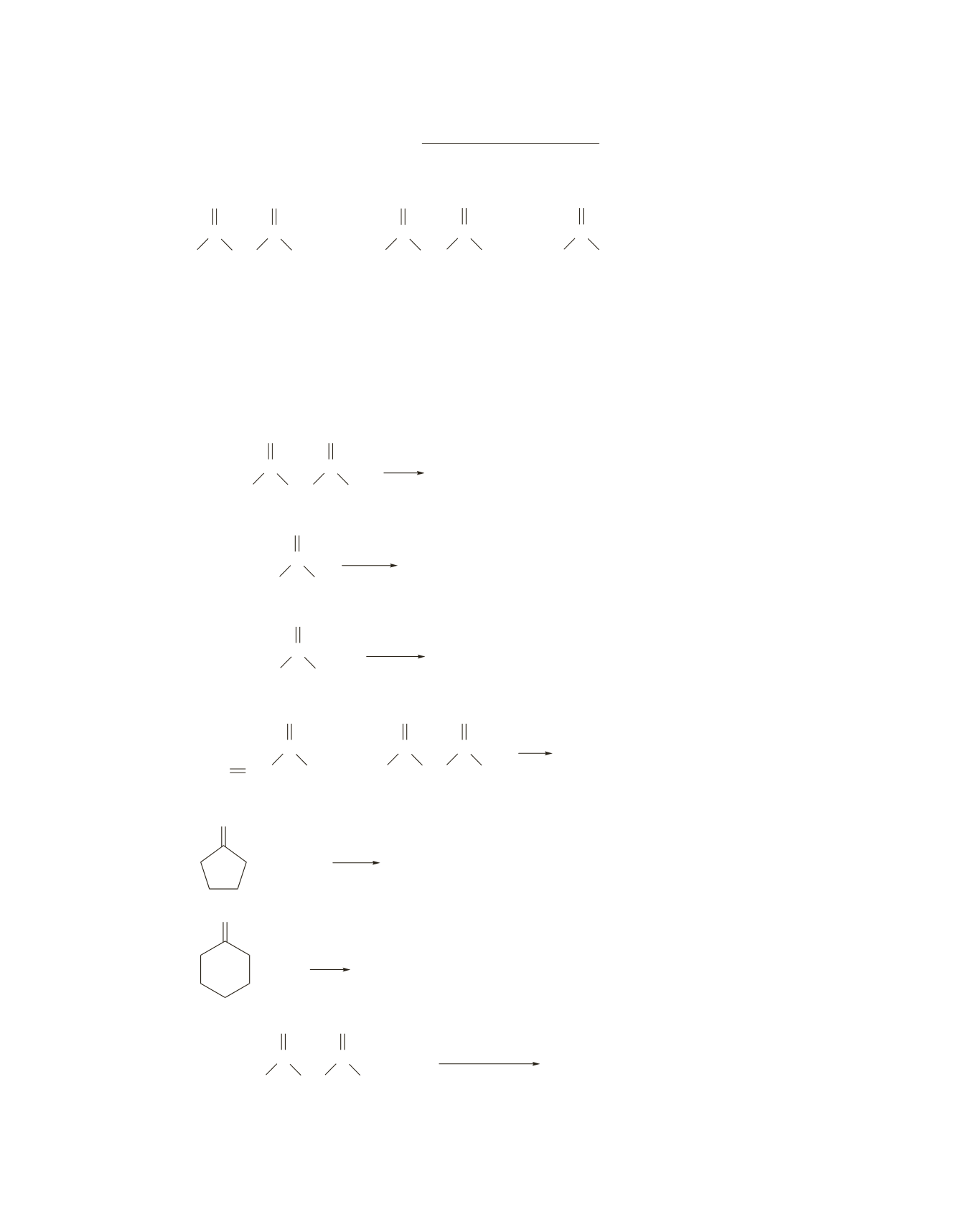
Chapter 17 595
Copyright © 2017 Pearson Education, Inc.
Chapter17 Practice Test
1.
Rank the following compounds from most acidic to least acidic:
CH
3
CH
3
OCH
3
CH
2
CH
3
CH
3
CH
2
CH
3
C
O
C
O
C
O
C
O
C
O
2.
Draw a structure for each of the following:
a.
the most stable enol tautomer of 2,4-pentanedione
b.
a
b
-keto ester
3.
Draw the product of the following reactions:
a.
CH
3
CH
2
CH
2
OH
C
O
C
O
b.
CH
3
CH
2
CH
2
H
Br
2
, HO
−
C
O
c.
CH
3
CH
2
CH
2
OCH
3
−
1. CH
3
O
2. HCl
C
O
d.
−
CH
2
CH CH
3
+
CH
3
CH
2
CH
3
HO
H
2
O
C
O
C
O
C
O
e.
O
Br
2
H
3
O
+
+
f.
HO
−
O
+
Cl
2
g.
CH
3
CH
2
O
2
OCH
CH
2
CH
3
−
1. CH
3
CH
2
O
2. CH
3
CH
2
CH
2
Br
3. HCl, H
2
O,
C
O
C
O
















