
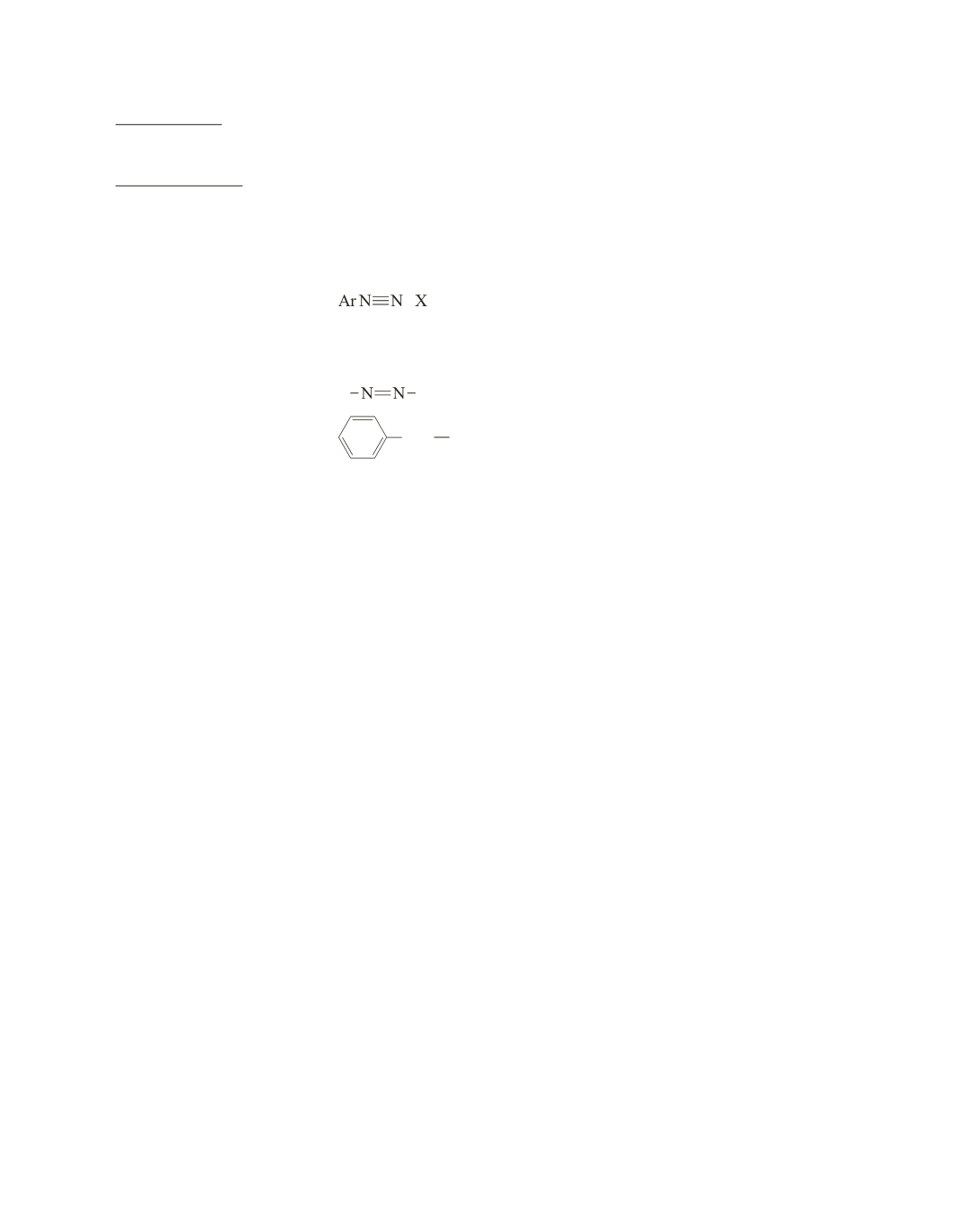
597
Copyright © 2017 Pearson Education, Inc.
CHAPTER
18
Reactions of Benzene and Substituted Benzenes
Important Terms
activating substituent
a substituent that increases the reactivity of an aromatic ring. Electron-donating
substituents activate aromatic rings toward electrophilic attack, and electron-withdrawing substituents activate aromatic rings toward nucleophilic attack.
arenediazonium salt
+
−
aromatic compound
a cyclic and planar compound with an uninterrupted cloud of electrons containing
an odd number of pairs of
p
electrons.
azo linkage
a
bond.
benzyl group
CH
2
deactivating substituent
a substituent that decreases the reactivity of an aromatic ring. Electron-withdrawing
substituents deactivate aromatic rings toward electrophilic attack, and electron-donating substituents deactivate aromatic rings toward nucleophilic attack.
donate electrons by
donation of electrons through
p
orbital overlap with neighboring
p
bonds.
resonance (resonance
electron donation)
electrophilic aromatic
a reaction in which an electrophile substitutes for a hydrogen of an aromatic ring.
substitution reaction
Friedel–Crafts acylation
an electrophilic aromatic substitution reaction that puts an acyl group on an
aromatic ring.
Friedel–Crafts alkylation
an electrophilic aromatic substitution reaction that puts an alkyl group on an
aromatic ring.
Gatterman–Koch reaction
a reaction that uses a high-pressure mixture of carbon monoxide and HCl to form
benzaldehyde.
halogenation
reaction with a halogen
1
Br
2
, Cl
2
, I
2
2
.
inductive electron
withdrawal of electrons through a
s
bond.
withdrawal
















