
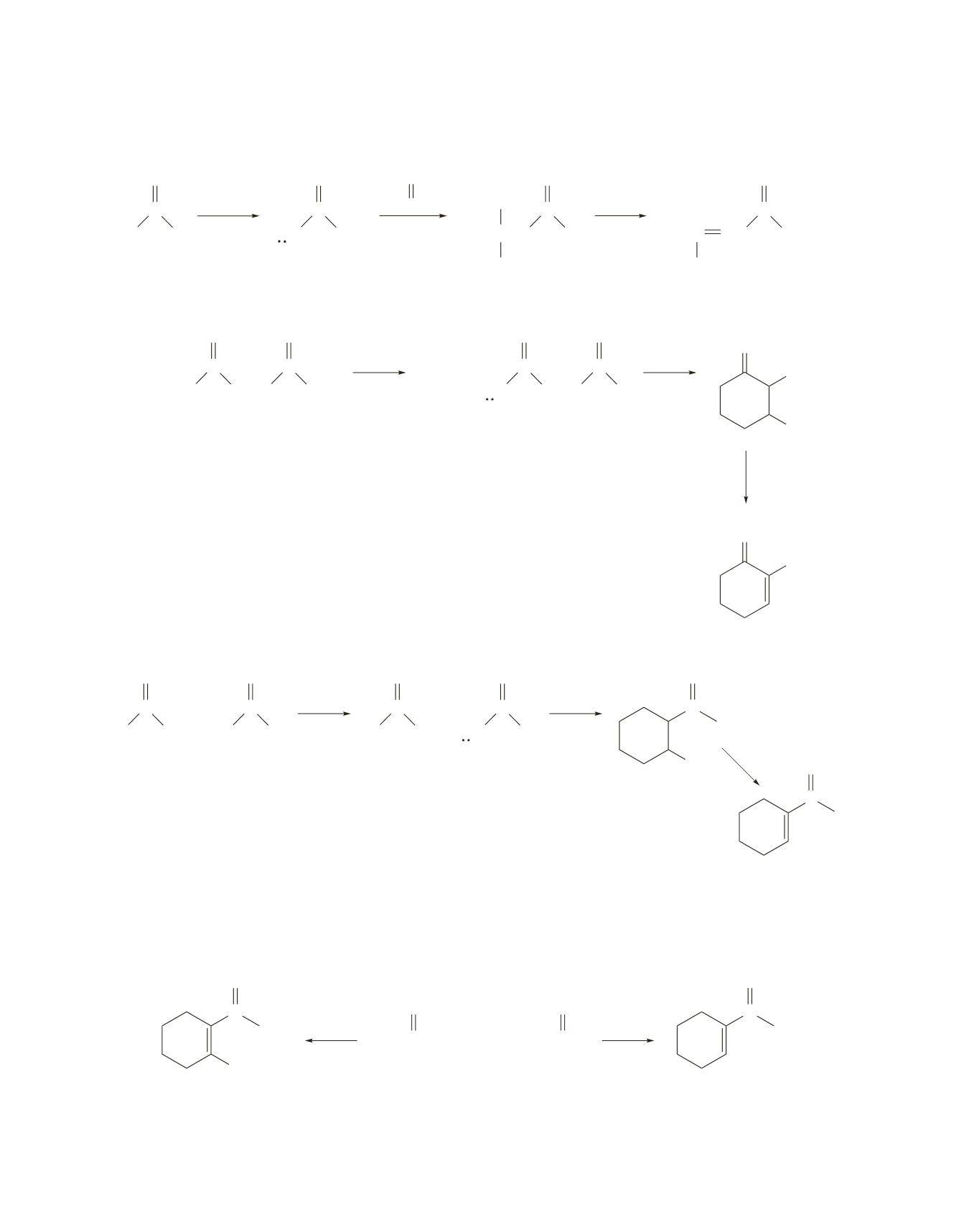
586 Chapter 17
Copyright © 2017 Pearson Education, Inc.
D
can be prepared by removing an
a
-hydrogen from acetaldehyde with LDA and then slowly adding
acetone, followed by dehydration.
CH
3
H
LDA/THF
CH
2
H
−
CH
3
CCH
3
O
add slowly
CH
3
CCH
2
H
O
CH
3
H
2
SO
4
CH
3
C CH H
CH
3
−
C
O
C
O
C
O
C
O
E
can be prepared via an intramolecular aldol condensation using 5-oxooctanal.
CH
3
CH
2
CH
2
(CH
2
)
3
(CH
2
)
3
H
CH
3
CH
2
CH
H
O
CH
2
CH
3
O
−
O
CH
2
CH
3
H
2
SO
4
−
C
O
C
O
C
O
C
O
HO
−
F
can be prepared via an intramolecular aldol condensation using 1,7-heptanedial.
C
H
O
−
−
O
C
H
O
H
2
SO
4
H (CH
2
)
4
CH
2
H
C
O
C
O
H (CH
2
)
4
CH H
C
O
C
O
HO
−
A small amount of
G
can be formed via an intramolecular aldol condensation using 7-oxooctanal, but this
compound can form two different
a
-carbanions that can react with a carbonyl group to form a six-mem-
bered ring. Because an aldehyde is more reactive than a ketone toward nucleophilic addition, the target
molecule will be a minor product.
CH
3
minor product
major product
C
H
O
C
CH
3
O
CH
3
CCH
2
CH
2
CH
2
CH
2
CH
2
CH
O
O
















