
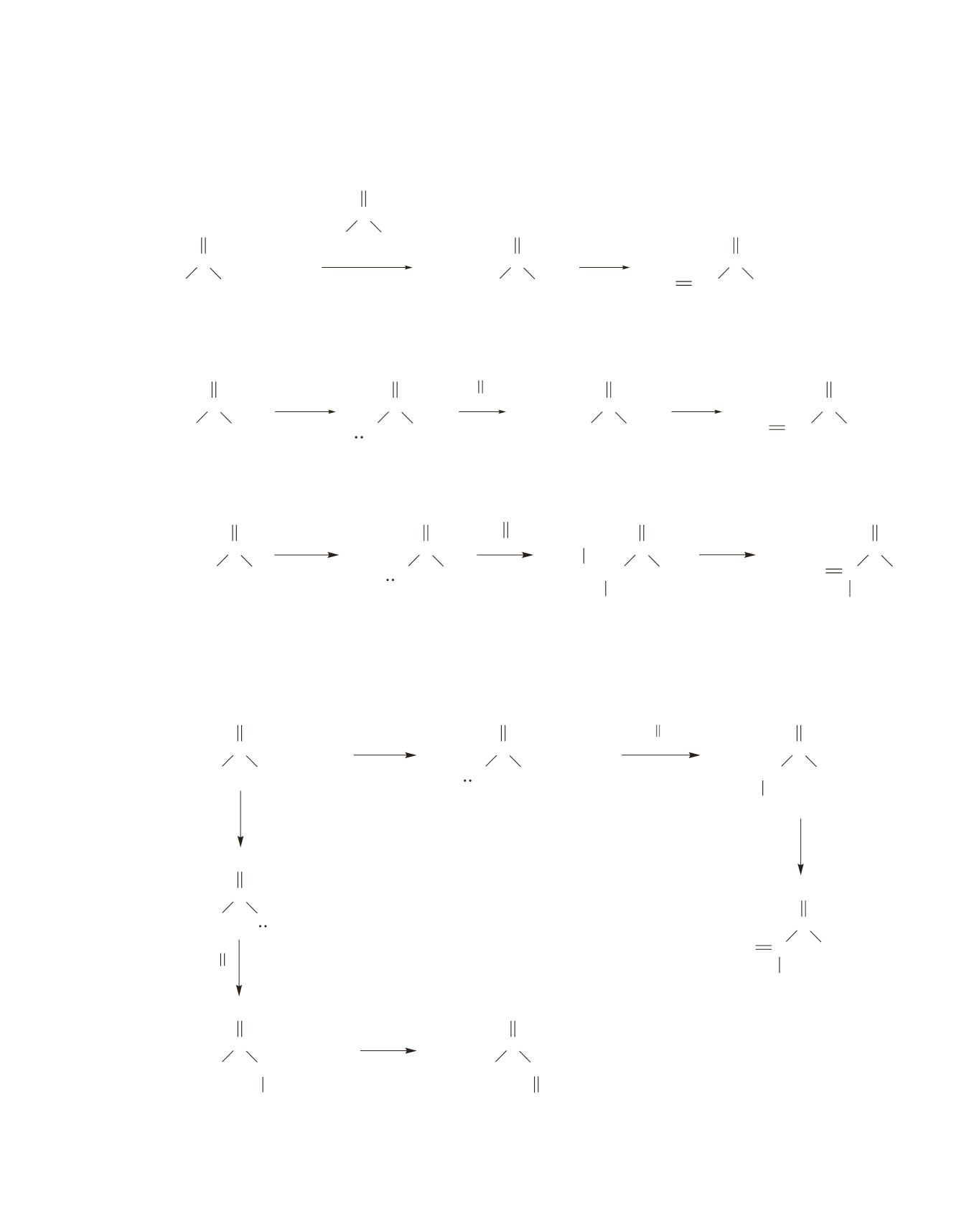
Chapter 17 585
Copyright © 2017 Pearson Education, Inc.
76.
A
can be prepared by adding the compound with
a
-hydrogens slowly to a basic solution of the compound
without
a
-hydrogens, followed by dehydration.
+
HO
H H
C
O
CH
3
CH
3
C
O
H
2
SO
4
−
−
OCH
2
CH
2
C
O
CH
3
CH
2
CH
C
O
CH
3
add slowly
Or
A
can be prepared by removing an
a
-hydrogen from acetone with LDA and then adding formaldehyde,
followed by dehydration.
CH
3
CH
3
LDA/THF
CH
2
CH
3
−
−
HCH
O
OCH
2
CH
2
H
2
SO
4
CH
2
CH
C
O
C
O
C
O
C
O
CH
3
CH
3
B
can be prepared by removing an
a
-hydrogen from propionaldehyde with LDA and then slowly adding
acetaldehyde, followed by dehydration.
CH
3
CH
2
H
LDA/THF
CH
3
CH H
−
−
CH
add slowly
3
CH
O
CH
3
CHCH H
H
O
CH
3
H
2
SO
4
CH
3
CH C
CH
3
C
O
C
O
C
O
C
O
C
can be prepared by removing an
a
-hydrogen from 3-hexanone with LDA and then adding formaldehyde,
followed by dehydration. The yield is poor because 3-hexanone is an asymmetrical ketone; therefore, two
different
a
-carbanions are formed that lead to two different
a
,
b
-unsaturated ketones.
CH
3
CH
2
CH
2
CH
2
CH
3
LDA/THF
CH
3
CH CH
2
CH
2
CH
3
HCH
O
OCH
2
CH CH
2
CH
2
CH
3
CH
3
−
−
H
2
SO
4
LDA/THF
HCH
O
CH
3
CH
2
CHCH
2
CH
3
−
−
CH
3
CH
2
CHCH
2
CH
3
CH
2
O
CH
3
CH
2
CCH
2
CH
3
CH
2
H
2
SO
4
C
2
CH CH
2
CH
3
CH
3
CH
2
C
O
C
O
C
O
C
O
C
O
C
O
C
O
















