
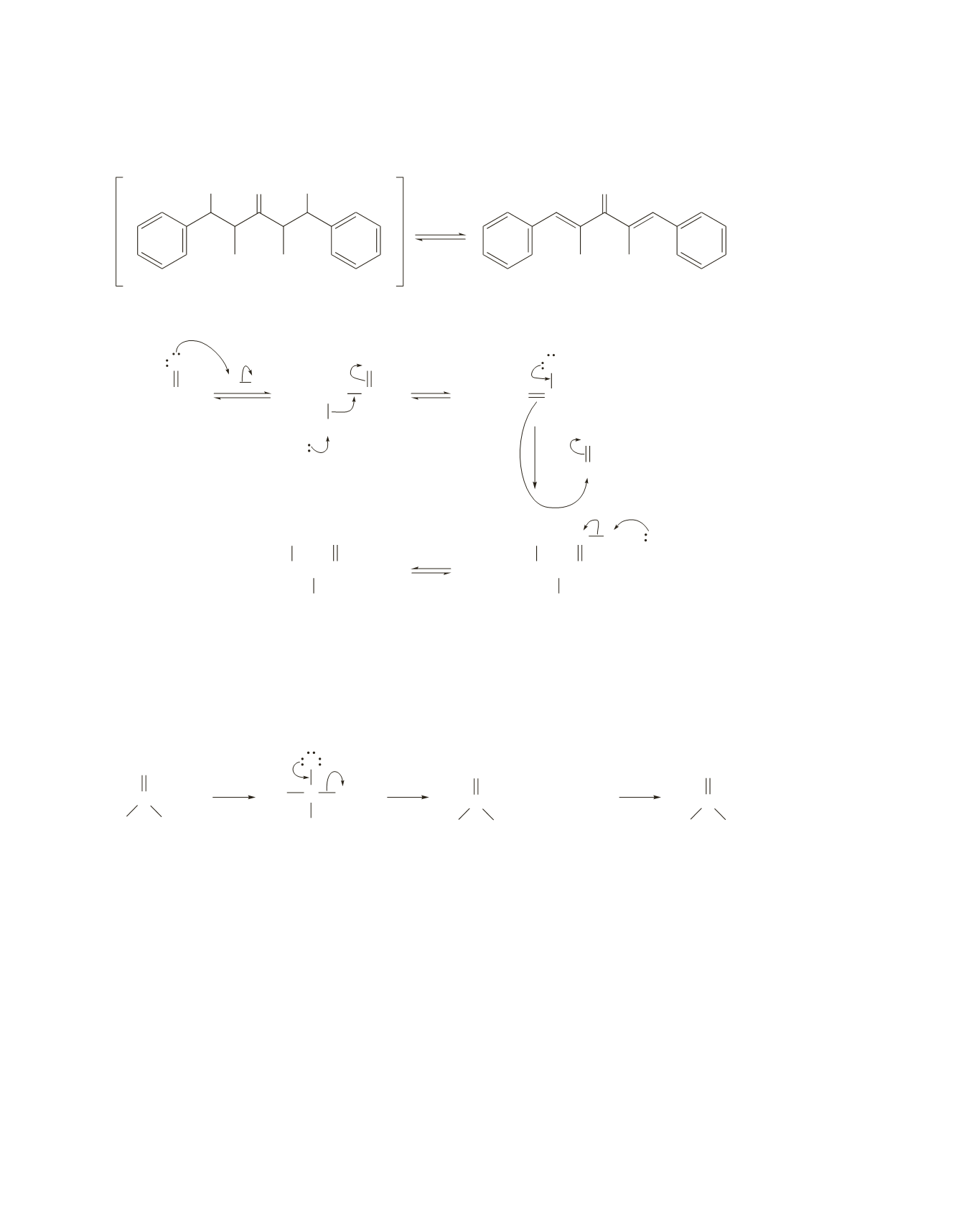
574 Chapter 17
Copyright © 2017 Pearson Education, Inc.
56.
Two aldol additions occur. The initially formed addition product loses water immediately because the new
double bonds are particularly stable (and therefore easy to form) since each is conjugated with a benzene ring.
O OH
OH
O
+
2 H
2
O
57.
CH
3
CH
2
CH
O
CH
3
CH CH
OH
H
B
CH
3
CH CH
OH
CH
3
CH
2
CH
OH
CH
3
CH
2
CHCHCH
OH H
CH
3
CH
3
CH
2
CHCHCH
OH
CH
3
HB
+
H B
+
HB
+
+
+
B
+
O
O
58.
The haloform reaction requires that a group be created that is a weaker base than hydroxide ion so that
hydroxide ion is not the group eliminated from the tetrahedral intermediate. For an alkyl group to be the
weaker base, the
a
-carbon must be bonded to three halogen atoms. The only alkyl group that can become
bonded to three halogen atoms at the
a
-carbon is a methyl group (because it is bonded to three hydrogens).
HO
−
−
CBr
3
R CBr
3
C
O
R OH
C
O
+
HCBr
3
R O
−
C
O
+
C R
CBr
3
OH
O
−
















