
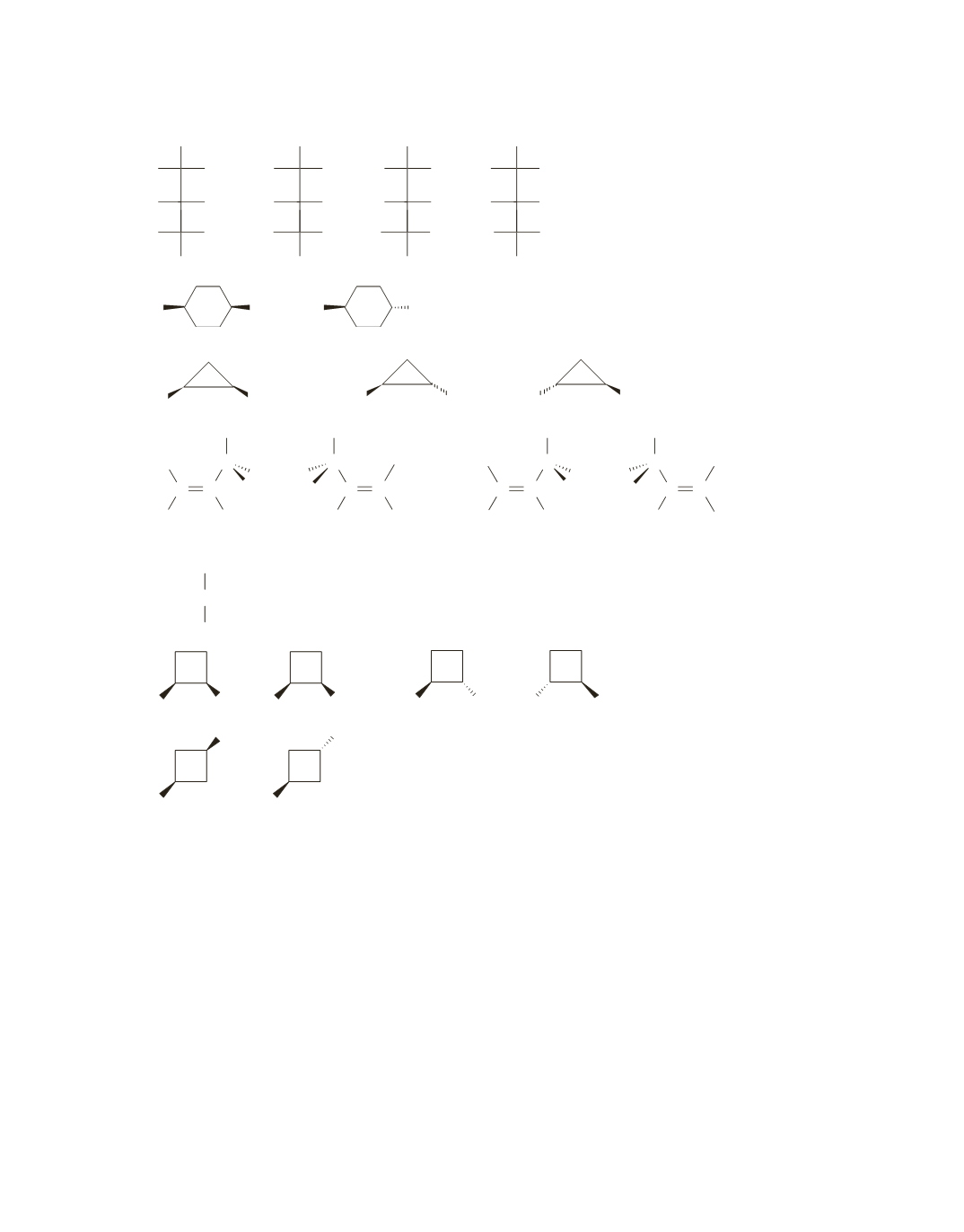
174 Chapter 4
Copyright © 2017 Pearson Education, Inc.
d.
CH
3
CH
3
H Cl
CH
3
CH
3
Cl
H
CH
3
CH
3
Cl
H
CH
3
CH
3
H Br
Br
H H Br Br
H
H H H H H H H H
H Cl
e.
Cl
Br
Br
Cl
cis and trans
f.
CH
3
CH
3
CH
3
3
CH
CH
3
CH
3
g.
C C
H
CH
3
C
H
CH
3
H
Br
CC
H
CH
3
CH
3
C
H
CH
3
H
Br
C C
H C
H
CH
3
H
Br
CC
CH
3
H C
H
CH
3
H
Br
R
S
R
S
cis
trans
h.
CH
3
CH
2
CCH
2
CH
3
CH
3
CH
3
No isomers are possible for this compound, because it does not have an
asymmetric center.
i.
Cl
Br
Cl
Br
Cl
Br
Br
Cl
j.
Cl
Br
Cl
Br
cis and trans only (no asymmetric centers)
67.
Only the fourth one
1
CHFBrCl
2
has an atom with four different atoms attached to a carbon, so it is the
only one that has an asymmetric center.
68.
a.
1
2
R
,3
R
2
-3-chloro-2-pentanol
b.
1
S
2
-2-methyl-1,2,5-pentanetriol
c.
1
2
S
,3
S
2
-1,2-dibromo-2-methyl-3-pentanol
















