
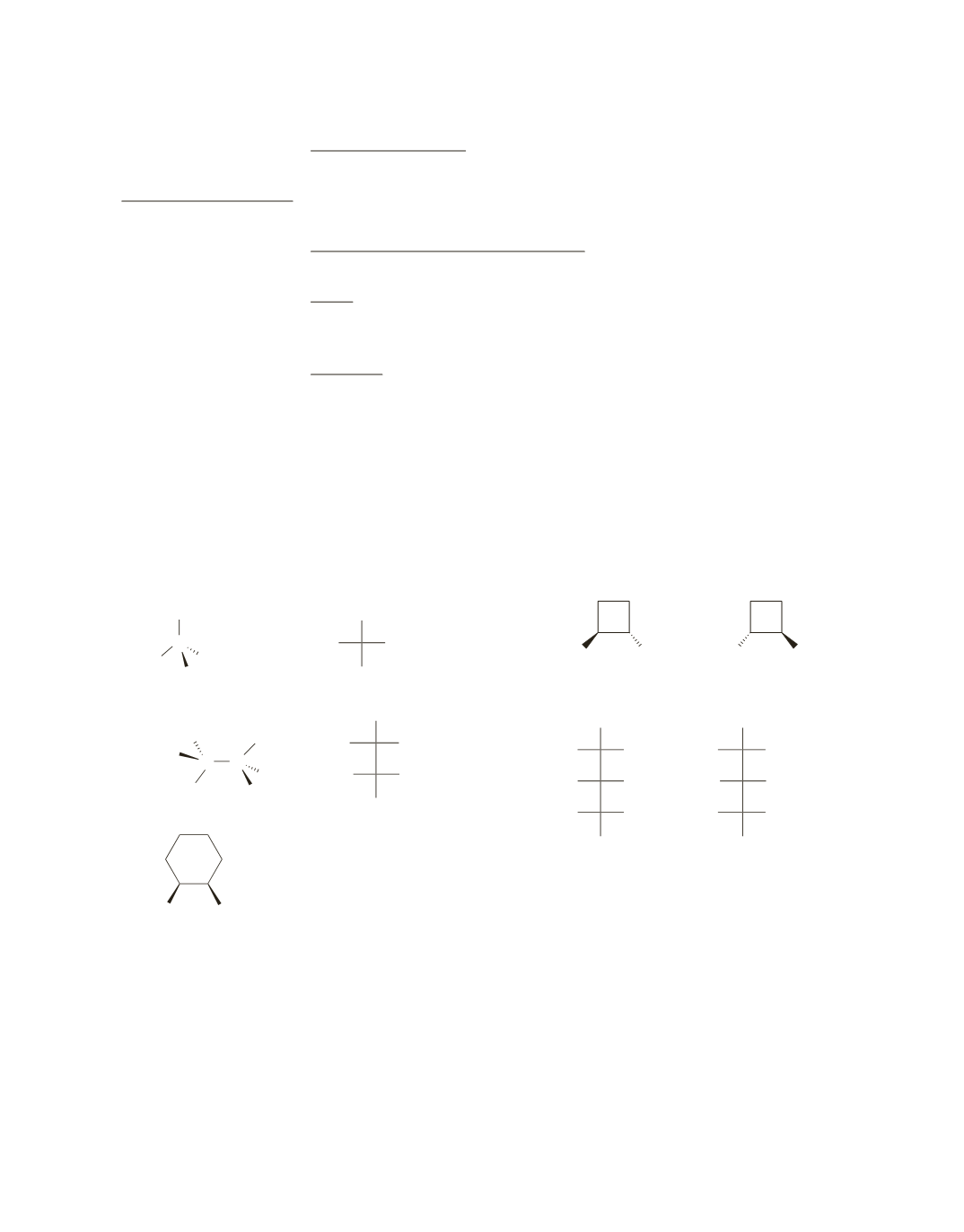
180 Chapter 4
Copyright © 2017 Pearson Education, Inc.
91.
observed speci c rotation
observed rotation
concentration
=
×
length
% optical purity
observed
−6.52°
0.187
g in100 mL
×
1 dm
=
= −
34 9.
speci c rotation
speci c rotation of the pure enantiomer
% of the (+)-isomer
×
=
=
=
100
34 9
39 0
100
89 5
100 89 5
2
5
−
−
×
− =
.
.
. %
.
. %
.
.
. %
25
89 5 5 25 94 75
% of the ( )-isomer
−
= + =
92.
a.
diastereomers [The configuration of all the symmetric centers is not the same in both (then the two
would be the same) and not opposite in both (then the two would be enantiomers.)] Recall that diaste-
reomers are stereoisomers that are not enantiomers.
b.
identical (by rotating the first compound clockwise, you can see that it is superimposable on the other)
c.
constitutional isomers
d.
diastereomers (the configuration of all the stereoisomers is not the same in both and not opposite in
both)
93.
a.
Br
C
H Cl
CH
2
CH
2
CH
3
Br
Cl
H
CH
2
CH
2
CH
3
or
d.
Br
Br
Br
Br
or
b.
CH
3
CH
2
C C
CH
3
H
Cl
H
Cl
CH
3
Cl
H
CH
2
CH
3
H Cl
or
e.
CH
2
CH
3
H CH
3
CH
3
H
CH
3
H
CH
2
CH
3
CH
2
CH
3
CH
3
H
CH
3
H
H
CH
3
CH
2
CH
3
c.
CH
3
CH
3
















