
Chapter 4 177
Copyright © 2017 Pearson Education, Inc.
83.
optical purity
=
+
1.4
+
8.7
=
0.16
=
16
,
excess
R
enantiomer
100
,
-
16
,
=
84
,
is a racemic mixture
R
enantiomer
=
1
>
2
1
84
,
2
+
16
,
=
42
,
+
16
,
=
58
,
84.
a.
R
c.
Cl
Cl
H
R
=
Cl
H CH
3
CH
2
CH
3
R
CH
S
e.
f.
b.
S
d.
Cl
=
H Cl
S
Cl
H CH
3
CH
2
CH
3
R
CH
3
H Cl
CH
2
CH
3
S
e.
f.
Fisher projections show the molecule with eclipsed bonds.
Therefore, to answer parts
e
and
f
, first rotate the Newman
projection so it is eclipsed. Then turn the Newman projection
into a Fisher projection. (See page 188 in the text.)
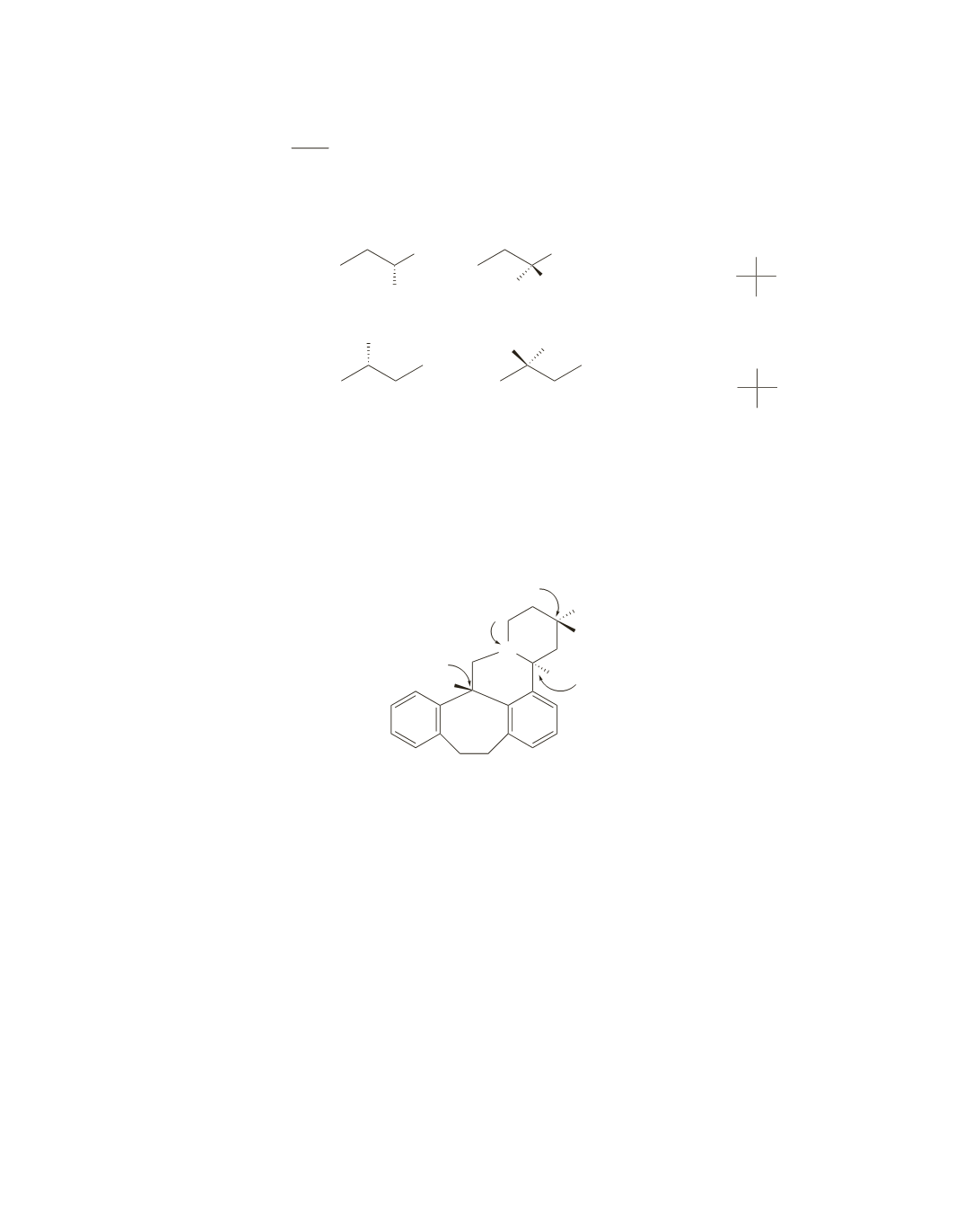
85.
Butaclamol has four asymmetric centers; three of them are carbons, and one is a nitrogen.
N
H
H
OH
C(CH
3
)
3
86.
The only way that
R
and
S
are related to
1
+
2
and
1
-
2
is that if the configuration of one enantiomer (for
example, the
R
enantiomer) is
1
-
2
, the the configuration of the other enantiomer is
1
+
2
.
Because some compounds with the
R
configuration are
1
+
2
and some are
1
-
2
, there is no way to determine
whether a particular
R
enantiomer is
1
+
2
or
1
-
2
without putting the compound in a polarimeter or finding
out whether someone else has previously determined how the compound rotates the plane of polarization
of plane-polarized light.
















