
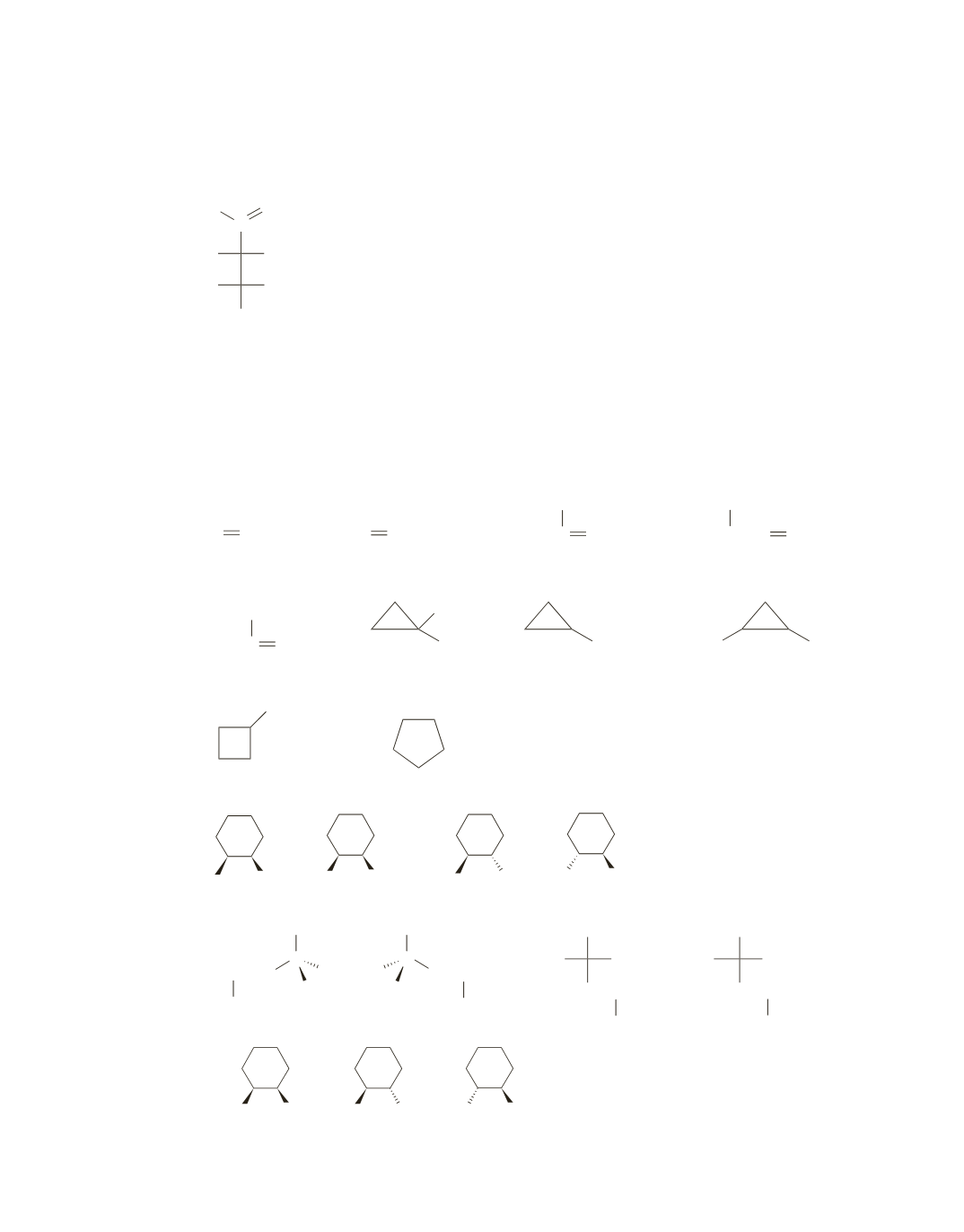
Chapter 4 173
Copyright © 2017 Pearson Education, Inc.
62.
Start by naming the first stereosiomer. Finding that
A
is 2
R
,3
R
allows you to answer both questions.
a. A
is d-erythrose.
C
OH
H
R
R
OH
H
CH
2
OH
O O
−
b.
d-Threose has the opposite configuration at C-2 and the same
configuration at C-3. Therefore,
C
is d-threose.
63.
Compound
A
has two stereoisomers, because it has an asymmetric center (at N).
Compound
B
does not have stereoisomers, because it does not have an asymmetric center.
Compound
C
has an asymmetric center at N but, because of the lone pair, the two enantiomers rapidly
interconvert, so it exists as a single compound.
64.
1
+
2
-Limonene has the
R
configuration, so it is the stereoisomer found in oranges and lemons.
65.
CH
3
CH CHCH
2
CH
3
2 stereoisomers
[cis and trans]
CH
2
CHCH
2
CH
2
CH
3
no stereoisomers
CH
3
C CHCH
3
no stereoisomers
CH
3
CH
3
CHCH CH
2
no stereoisomers
CH
3
CH
3
CH
2
C CH
2
no stereoisomers
CH
3
CH
3
CH
3
no stereoisomers
no stereoisomers
3 stereoisomers
[Cis is a meso compound.]
[Trans is a pair of enantiomers.]
CH
3
no stereoisomers
no stereoisomers
CH
2
CH
3
CH
3
CH
3
66.
a.
Br
Cl
Cl
Br
Br
Cl
Br
Cl
b.
Br
C
CH
3
CHCH
2
H
CH
3
CH
3
CH
3
Br
C
CH
2
CHCH
3
H
CH
3
CH
2
CHCH
3
Br
H
CH
3
CH
3
CH
2
CHCH
3
Br
H
CH
3
+
or
+
CH
3
c.
Cl
Cl
Cl
Cl
Cl
Cl
a meso compound
















