
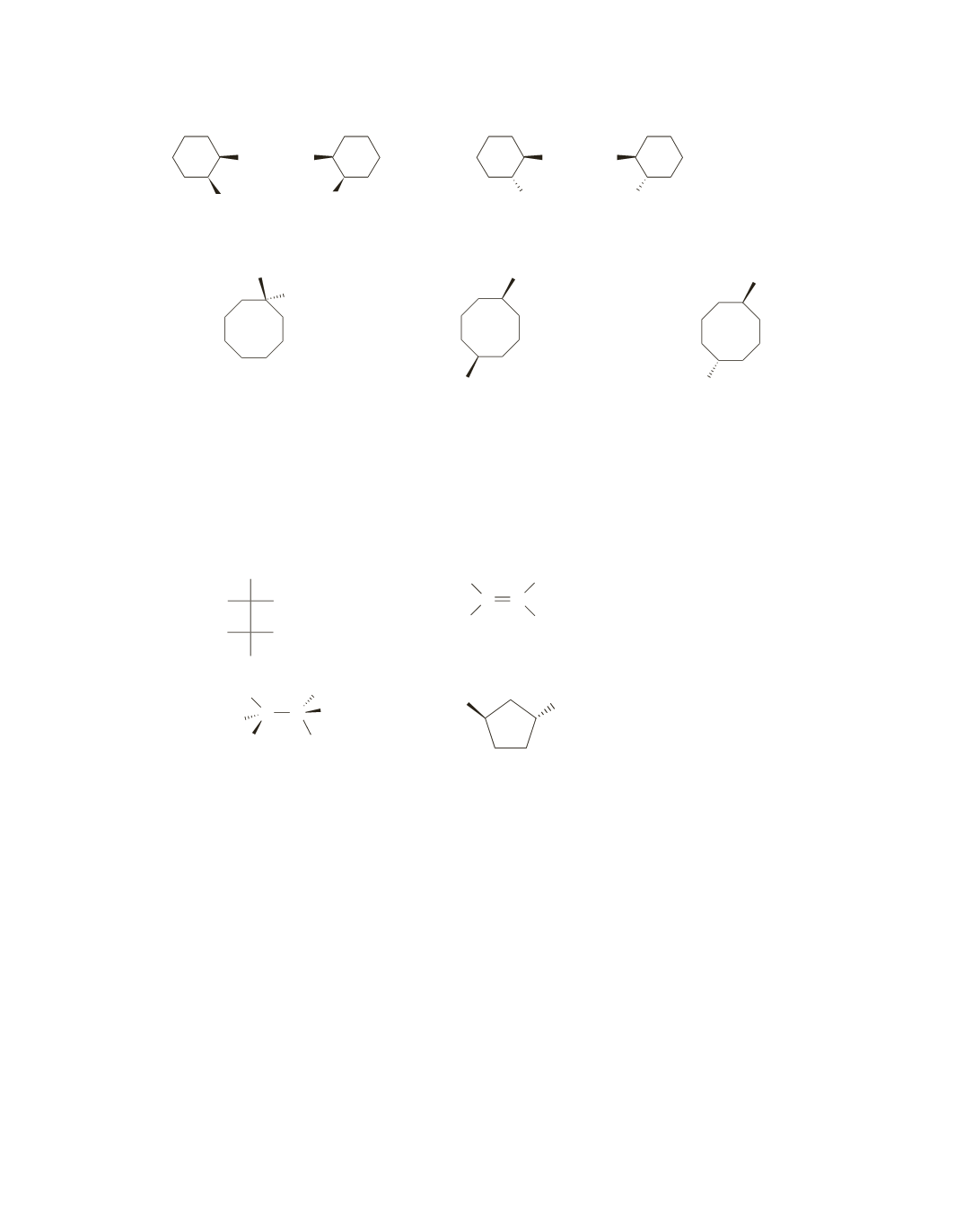
Chapter 4 169
Copyright © 2017 Pearson Education, Inc.
45.
OH HO
CH
3
CH
3
CH
3
R
S
R
S
OH HO
CH
3
R
R
S
S
cis
-2-methylcyclohexanol
trans
-2-methylcyclohexanol
46.
CH
3
Cl
1-chloro-1-methylcyclooctane
cis
-1-chloro-5-methylcyclooctane
trans
-1-chloro-5-methylcyclooctane
CH
3
Cl
CH
3
Cl
47.
There is more than one diastereomer for
a, b,
and
d
;
c
has only one diastereomer.
To draw a diastereomer, switch any one pair of substituents bonded to one of the asymmetric centers.
Because any one pair can be switched, your diastereomer may not be the one drawn here, unless you hap-
pened to switch the same pair that was switched here.
a.
CH
3
OH
H
H
HO
CH
3
b.
Cl
CH
3
CH
2
C C Cl
CH
3
H
H
c.
C C
H
CH
3
H
H
3
C
d.
HO
CH
3
48.
A
=
identical
B
=
enantiomer
C
=
diastereomer
D
=
identical
49.
B
,
D
, and
F
because each has two asymmetric centers and the same four groups bonded to each of the
asymmetric centers.
A
has two asymmetric centers, but it does not have a stereoisomer that is a meso compound, because it
does not have the same four groups bonded to each of the asymmetric centers.
C
and
E
do not have a stereoisomer that is a meso compound, because they do not have asymmetric
centers.
50.
Solved in the text.
















