
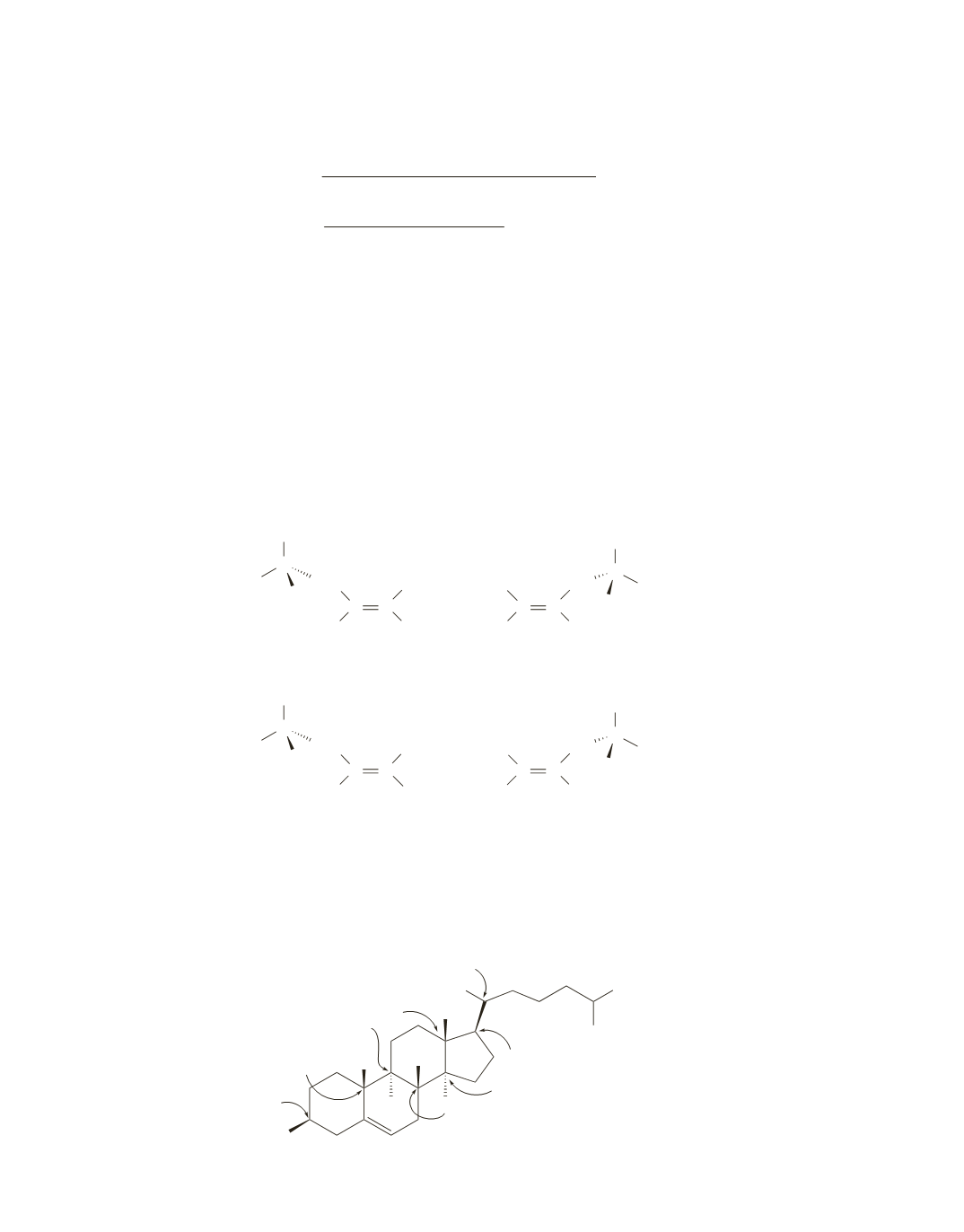
166 Chapter 4
Copyright © 2017 Pearson Education, Inc.
b.
50% of the mixture is excess ( )-mandelic acid.
optical puri
+
ty
observed speci c rotation
speci c rotation of th
= =
0 50.
e pure enantiomer
observed speci c rotation
obser
0 50
158
.
=
+
ved speci c rotation
= +
79
c.
50% of the mixture is excess
1
-
2
-mandelic acid.
observed specific
rotation
=
-
79
(For the calculation, see part
b
.)
36.
a.
From the data given, you cannot determine the configuration of naproxen.
b.
97% of the commercial preparation is
1
+
2
-naproxen; 3% is a racemic mixture. Therefore, the
commercial preparation forms 98.5%
1
+
2
-naproxen and 1.5%
1
-
2
-naproxen.
37.
Solved in the text.
38.
As a result of the double bond, the compound has a cis isomer and a trans isomer. Because the compound
also has an asymmetric center, the cis isomer can exist as a pair of enantiomers and the trans isomer can
exist as a pair of enantiomers.
C C
H
CH
3
H
CH
2
C
Br
CH
2
CH
3
H
CC
H
H
3
C
H
CH
2
C
Br
CH
2
CH
3
H
cis enantiomers
C C
CH
3
CH
3
H
H
CH
2
C
Br
CH
2
CH
3
H
CC
H
H
CH
2
C
Br
CH
2
CH
3
H
trans enantiomers
39.
a.
enantiomers
b.
identical compounds (Therefore, they are not stereoisomers.)
c.
diastereomers
40.
a.
Find the
sp
3
carbons that are bonded to four different substituents; these are the asymmetric centers.
Cholesterol has eight asymmetric centers. They are indicated by arrows.
CH
3
CH
3
HO
H
3
C
H
3
C
CH
3
H
H H
















