
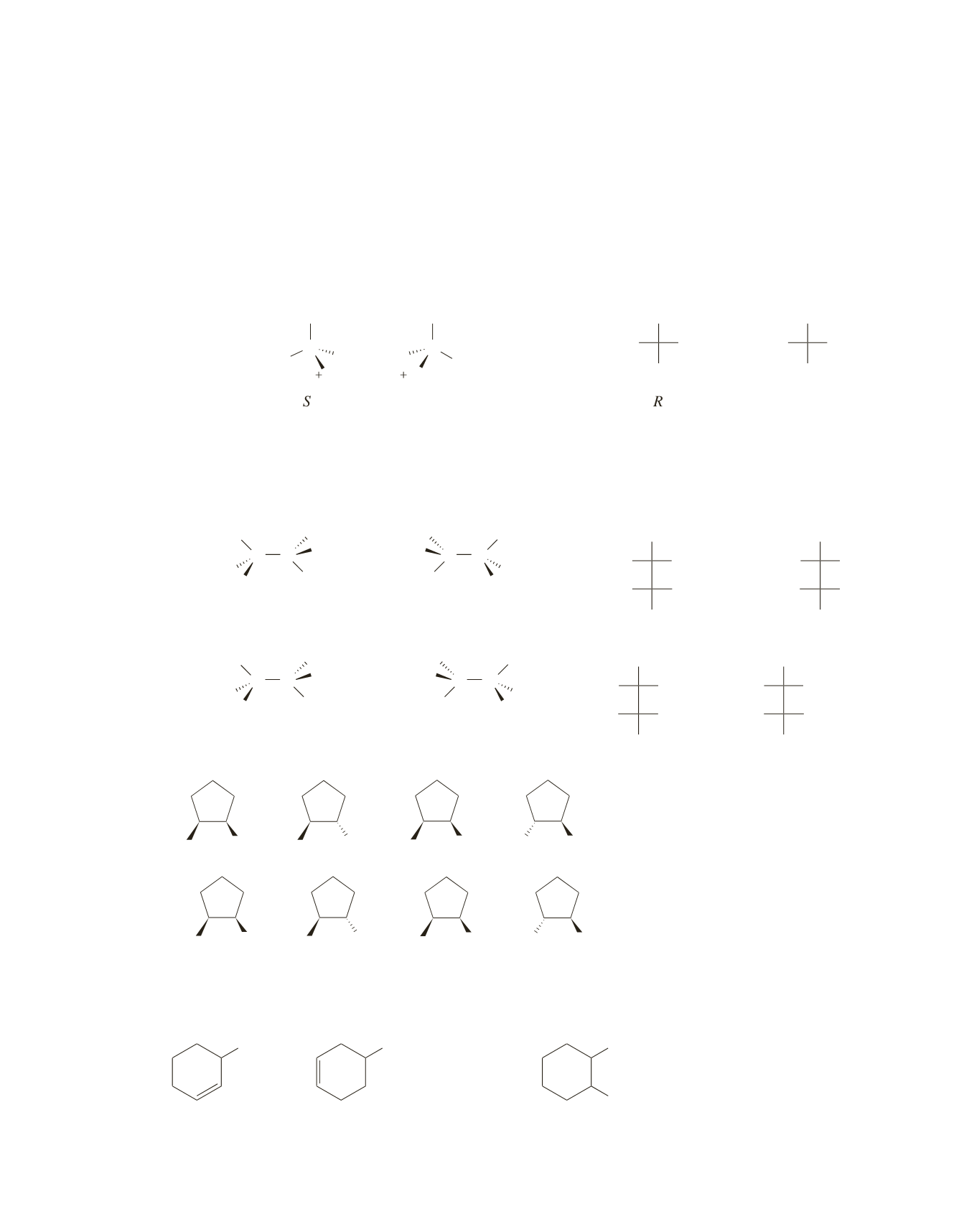
Chapter 4 167
Copyright © 2017 Pearson Education, Inc.
b.
2
8
=
256
Only the stereoisomer shown above is found in nature.
41.
Your perspective formulas may not look exactly like the ones drawn here because you can draw the first
one with the groups attached to any bonds you want. Just make certain that the second one is a mirror
image of the first one.
a.
Leucine has one asymmetric center, so it has two stereoisomers.
(CH
3
)
2
CHCH
2
C
COO
−
H
NH
3
C
COO
−
H
NH
3
CH
2
CH(CH
3
)
2
COO
−
NH
3
CH
2
CH(CH
3
)
2
H
+
COO
−
H
3
N
CH
2
CH(CH
3
)
2
H
+
or
R
S
b.
Isoleucine has two asymmetric centers, so it has four stereoisomers. Again, your perspective formulas
may not look like the ones drawn here. To make sure you have all four, determine the configuration
of each of the asymmetric centers. You should have
R
,
R
,
S
,
S
,
R
,
S
, and
S
,
R
. Notice that the asymmetric
centers in mirror images have the opposite configurations.
C
−
OOC
H
3
N
H
C
H
CH
2
CH
3
CH
3
C
COO
−
NH
3
H
C
H
H
3
C
+
+
COO
−
NH
3
H
+
CH
3
CH
3
H
CH
2
CH
3
COO
−
H
3
N H
+
H
CH
2
CH
3
or
CH
3
CH
2
S S
R R
R
R
S
S
CH
3
CH
3
CH
2
C
COO
−
H
NH
3
C
H
H
3
C
+
C
−
OOC
H
H
3
N
C
H
CH
2
CH
3
CH
3
+
COO
−
H
3
N H
+
H CH
3
CH
2
CH
3
COO
−
NH
3
H
+
H
CH
2
CH
3
R S
R S
R
S
S
R
42.
CH
3
Br
and
CH
3
Br
CH
3
CH
3
Br
Br
and
Br
CH
3
CH
3
Br
and
CH
3
CH
3
Br
Br
and
43.
B
and
D
have no symmetric centers.
A
and
C
each has one asymmetric center.
E
has two asymmetric centers.
*
A
*
C
*
*
E
















