
Chapter 4 165
Copyright © 2017 Pearson Education, Inc.
22.
a.
R
b.
To determine the configuration, first add the fourth bond to the asymmetric center. Remember that it
cannot be drawn between the two solid bonds. (It can be drawn on either side of the solid wedge.)
R
H OH
c.
R
.
23.
a.
S
b.
R
c.
S
d.
S
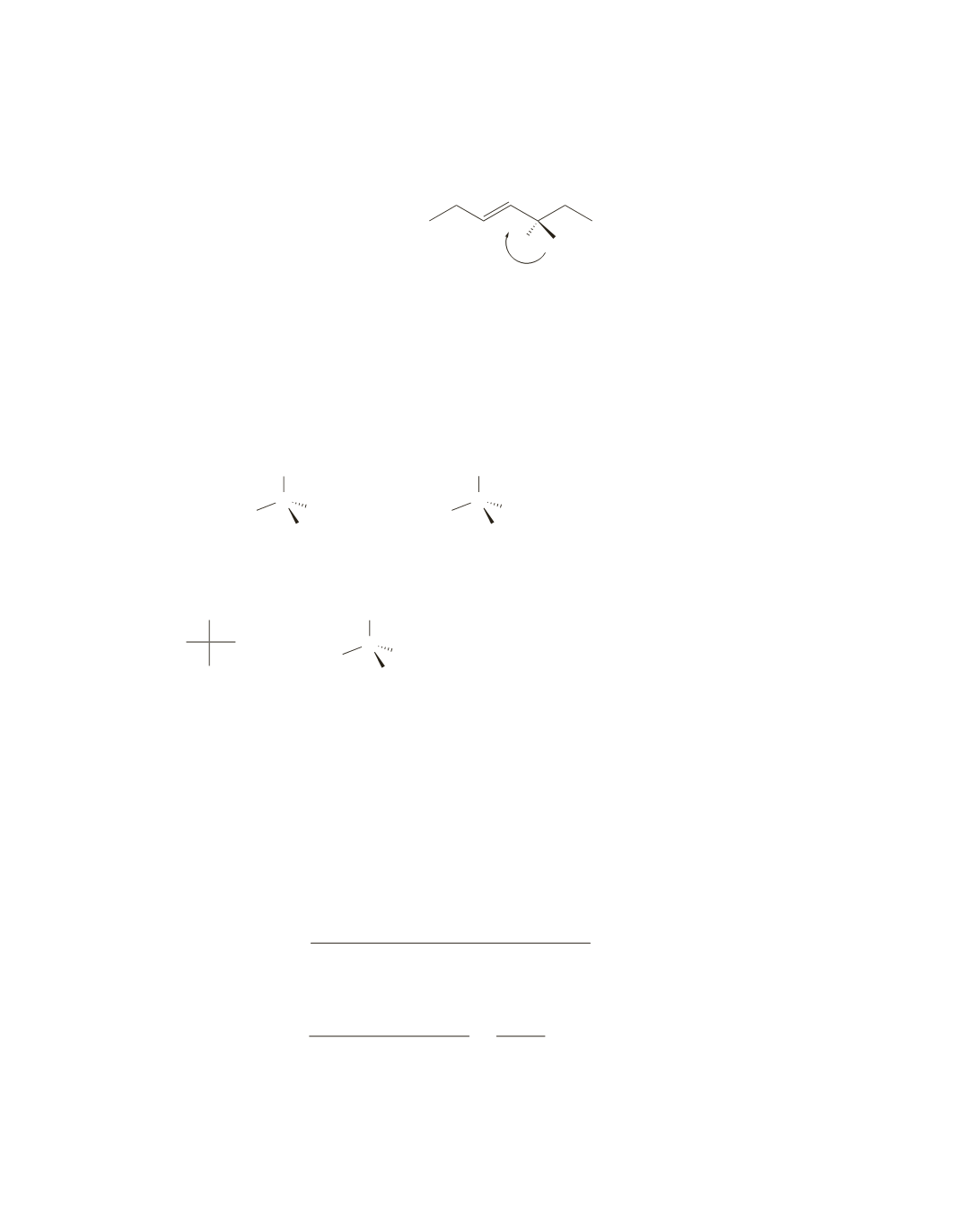
24.
The easiest way to determine whether two compounds are identical or enantiomers is to determine
their configurations: if both are
R
(or both are
S
), they are identical, one is
R
and the other is
S
, they are
enantiomers.
a.
identical
b.
enantiomers
c.
enantiomers
d.
enantiomers
25.
CH
3
CH
2
C
Cl
H
CH
3
a.
b.
CH
3
CH
2
C
Br
H
CH
2
Br
26.
Solved in the text.
27.
CH
3
CH
2
CH
3
H
COO
−
CH
2
CH
3
C
H
CH
3
COO
−
28.
a.
levorotatory
b.
dextrorotatory
29.
Solved in the text.
30.
a.
R
b.
R
c.
S
31.
Solved in the text.
32.
We see that the
1
R
2
-
alkyl halide reacts with HO
-
to form the
1
R
2
-alcohol. We are told that the product (the
1
R
2
-alcohol) is
1
-
2
. We can, therefore, conclude that the
1
+
2
-alcohol has the
S
configuration.
33.
speci c rotation
observed rotation (degrees)
=
[
+13.4
]
=
+13.4°
=
° = +
4
×
2
8
1.68
concentration (g in 100 mL)
×
length (dm)
There are 4 g in 100 mL, so
34.
a.
-
24
b.
0
35.
a.
0 (It is a racemic mixture.)
















