
244 Chapter 7
Copyright © 2017 Pearson Education, Inc.
c.
1-methoxy-2-pentyne
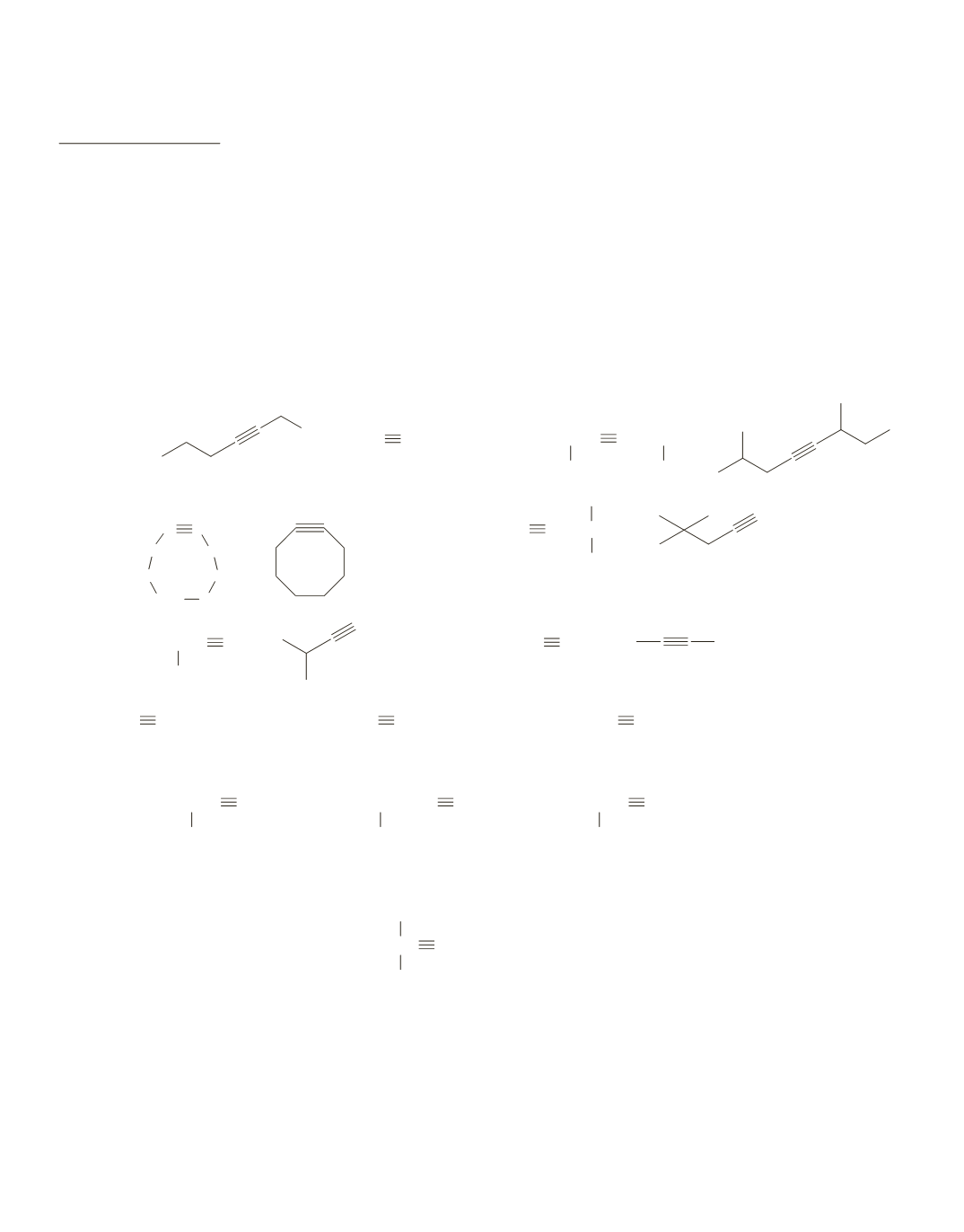
d.
3-ethyl-1-hexyne
Solutions to Problems
1.
a.
5-bromo-2-pentyne
b.
6-bromo-2-chloro-4-octyne
2.
a.
6-methyl-2-octyne
b.
5-ethyl-4-methyl-1-heptyne
c.
2-bromo-4-octyne
3.
The general molecular formula of a noncyclic hydrocarbon is
C
n
H
2
n
+
2
.
Therefore, the molecular formula for a noncyclic hydrocarbon with 14 carbons is
C
14
H
30
.
Because a compound has two fewer hydrogens for every ring and
p
bond, a compound with one ring and 4
p
bonds (2 triple bonds) has 10 fewer hydrogens than the C
n
H
2
n
+
2
formula.
Therefore, the molecular formula is
C
14
H
20
.
4.
a.
C1
ClCH
2
CH
2
C CCH
2
CH
3
b.
C C
CH
2
CH
2
CH
2
CH
2
CH
2
CH
2
c.
CH
3
CHC
CH
3
CH
d.
CH
3
CH
2
CHC CCH
2
CHCH
3
CH
3
CH
3
e.
HC CCH
2
CCH
3
CH
3
CH
3
f.
CH
3
C CCH
3
5.
HC CCH
2
CH
2
CH
2
CH
3
CH
3
C CCH
2
CH
2
CH
3
CH
3
CH
2
C CCH
2
CH
3
1-hexyne
butylacetylene
2-hexyne
methylpropylacetylene
3-hexyne
diethylacetylene
CH
3
CH
2
CHC CH
CH
3
CHCH
2
C CH
CH
3
CHC CCH
3
3-methyl-1-pentyne
sec
-butylacetylene
4-methyl-1-pentyne
isobutylacetylene
CH
3
CH
3
CH
3
4-methyl-2-pentyne
isopropylmethylacetylene
CH
3
CC
3,3-dimethyl-1-butyne
tert
-butylacetylene
CH
CH
3
CH
3
6.
a.
1-hepten-4-yne
b.
4-methyl-1,4-hexadiene
c.
5-vinyl-5-octen-1-yne
(One of the functional groups
cannot be included in the
parent hydrocarbon.)
d.
3-butyn-1-ol
e.
1,3,5-heptatriene
f.
2,4-dimethyl-4-hexen-1-ol
















