
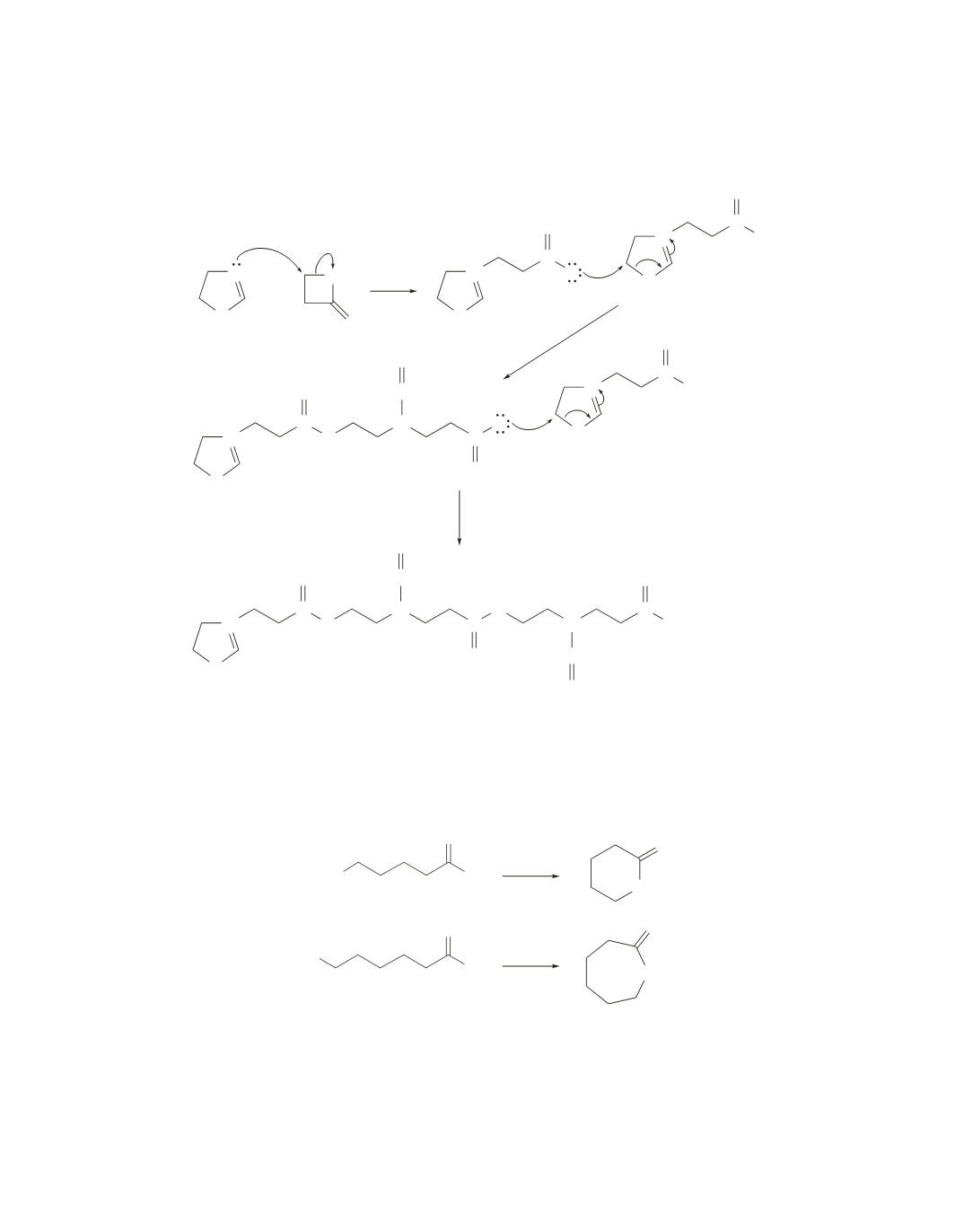
842 Chapter 27
Copyright © 2017 Pearson Education, Inc.
39.
The five-membered ring and the four-membered ring react to form the species that polymerizes to form the
alternating copolymer.
N
O
O
O
N
O
+
C
O
O
−
N
+
C
O
O
−
O
N
O
+
C
O
O
O
N
+
C
O
O
−
N
C
O
−
O
CH
O
N
O
+
C
O
O
N
C
O
O
CH
O
N
C
O
CH
O
O
−
40.
Both compounds can form esters via intramolecular or intermolecular reactions. The product of the
intramolecular reaction is a lactone; the intermolecular reaction leads to a polymer.
5-Hydroxypentanoic acid reacts intramolecularly to form a six-membered-ring lactone, whereas
6-hydroxyhexanoic acid reacts intramolecularly to form a seven-membered-ring lactone.
5-hydroxypentanoic acid
6-hydroxyhexanoic acid
O
O
O
O
HO
OH
O
OH
O
HO
The compound that forms the most polymer is the one that forms the least lactone because the two reac-
tions compete with each other.
The six-membered-ring lactone is more stable and, therefore, has a more stable transition state for its for-
mation, compared to a seven-membered-ring lactone. Because it is easier for 5-hydroxypentanoic acid to
form the six-membered-ring lactone than for 6-hydroxyhexanoic acid to form the seven-membered-ring
lactone,
6-hydroxyhexanoic acid
forms more polymer.
















