
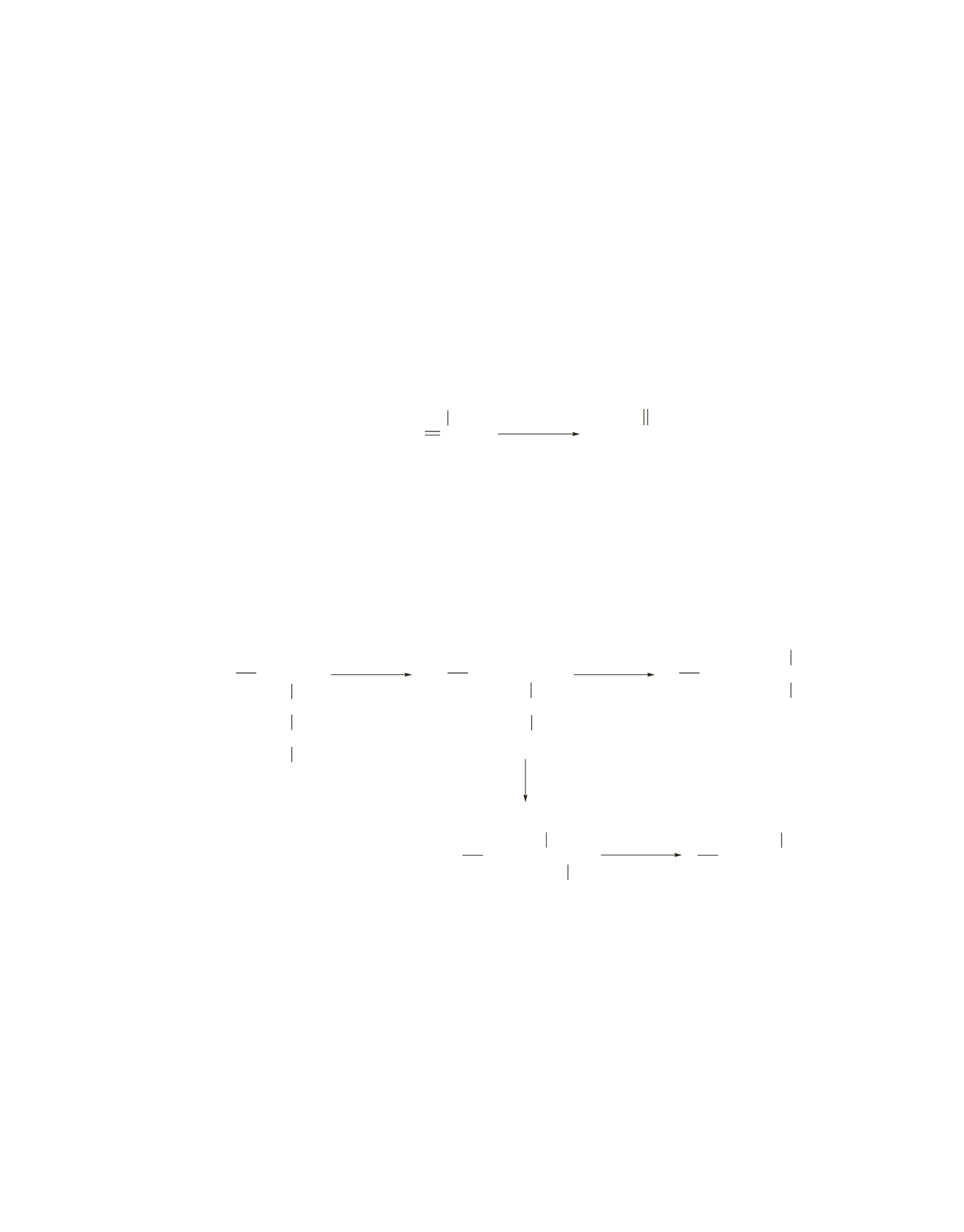
Chapter 27 839
Copyright © 2017 Pearson Education, Inc.
33.
The polymer in the flask that contained a high-molecular-weight polymer and little material of intermedi-
ate molecular weight was formed by a chain-growth mechanism, whereas the polymer in the flask that
contained mainly material of intermediate molecular weight was formed by a step-growth mechanism.
In a chain-growth mechanism, monomers are added to the growing end of a chain. This means that at any
one time, there will be polymeric chains and monomers.
Step-growth polymerization is not a chain reaction; any two monomers can react. Therefore, high-molecular-weight material will not be formed until the end of the reaction when pieces of intermediate
molecular weight combine.
34.
a.
Vinyl alcohol is unstable; it tautomerizes to acetaldehyde.
CH
2
CH
OH
vinyl alchol
tautomerization
CH
3
CH
acetaldehyde
O
b.
It is not a true polyester. It has ester groups as substituents
on
the backbone of the chain, but it does not
have ester groups
within
the backbone of the polymer chain. A true polyester has ester groups within
the backbone of the polymer chain.
35.
Each of the following five carbocations can add the growing end of the polymer chain.
CH
2
CH
CH
2
CHCH
3
CH
3
+
1,2-hydride
shift
CH
2
CH
2
CH
+
CHCH
3
CH
3
1,2-hydride
shift
CH
2
CH
2
CH
2
C
+
CH
3
CH
3
1,2-methyl
shift
CH
2
CH
2
CHCH
+
CH
3
CH
3
1,2-hydride
shift
CH
2
CH
2
CCH
2
CH
3
+
CH
3
















