
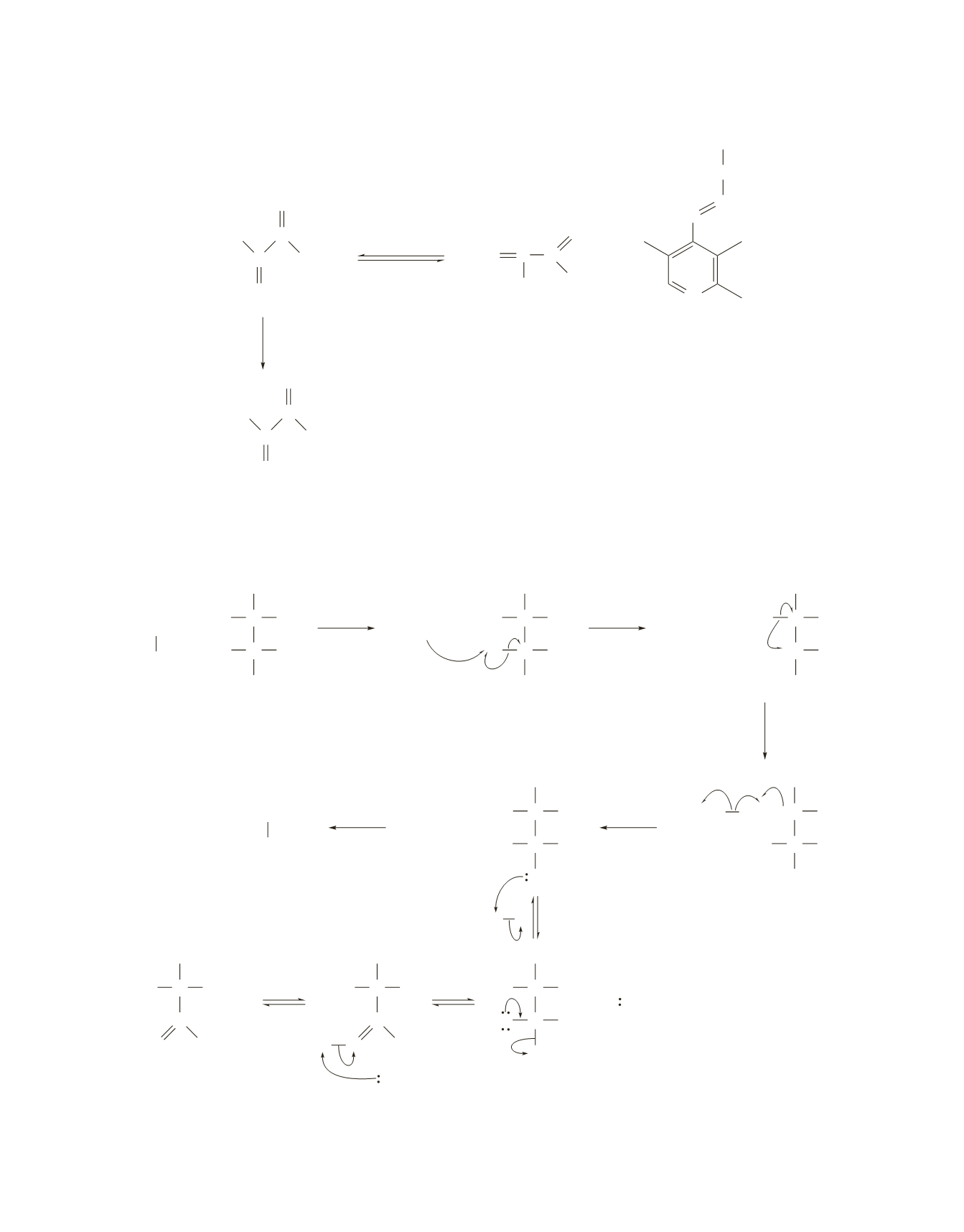
764 Chapter 23
Copyright © 2017 Pearson Education, Inc.
+
NH
4
+
H
2
O
HCl
tautomerization
_
CH
3
NH
+
(CH
2
)
4
N
E
C
NH
2
CH
2
(CH
2
)
4
NH
2
E
transimination
imine
hydrolysis
H
N
H
+
C
O
_
O
O C
C
O
_
CH
3
O
O C
C
O
HC
an enamine
19.
The mechanism is the same as that shown in the text for dioldehydrase. The tetrahedral intermediate that
is formed as a result of the coenzyme
B
12
–catalyzed isomerization is unstable and loses ammonia to give
acetaldehyde, the final product of the reaction.
H
C HO H
C
NH
2
H H
AdCH
2
Co(III)
H
C HO H
C
NH
2
H H
AdCH
2
Co(II)
H
C HO H
C
NH
2
H
AdCH
3
Co(II)
•
H
CH H
C HO H
AdCH
2
Co(II)
H
C H
C
NH
2
HO H
AdCH
2
Co(II)
•
•
H
•
AdCH
2
Co(II)
H
CH
BH
H
C HO H
B
NH
3
H
CH H
O H NH
4
+
+
+
+
+
NH
3
+
NH
2
C
H
CH
H
H
O H
C
















