
Chapter 23 763
Copyright © 2017 Pearson Education, Inc.
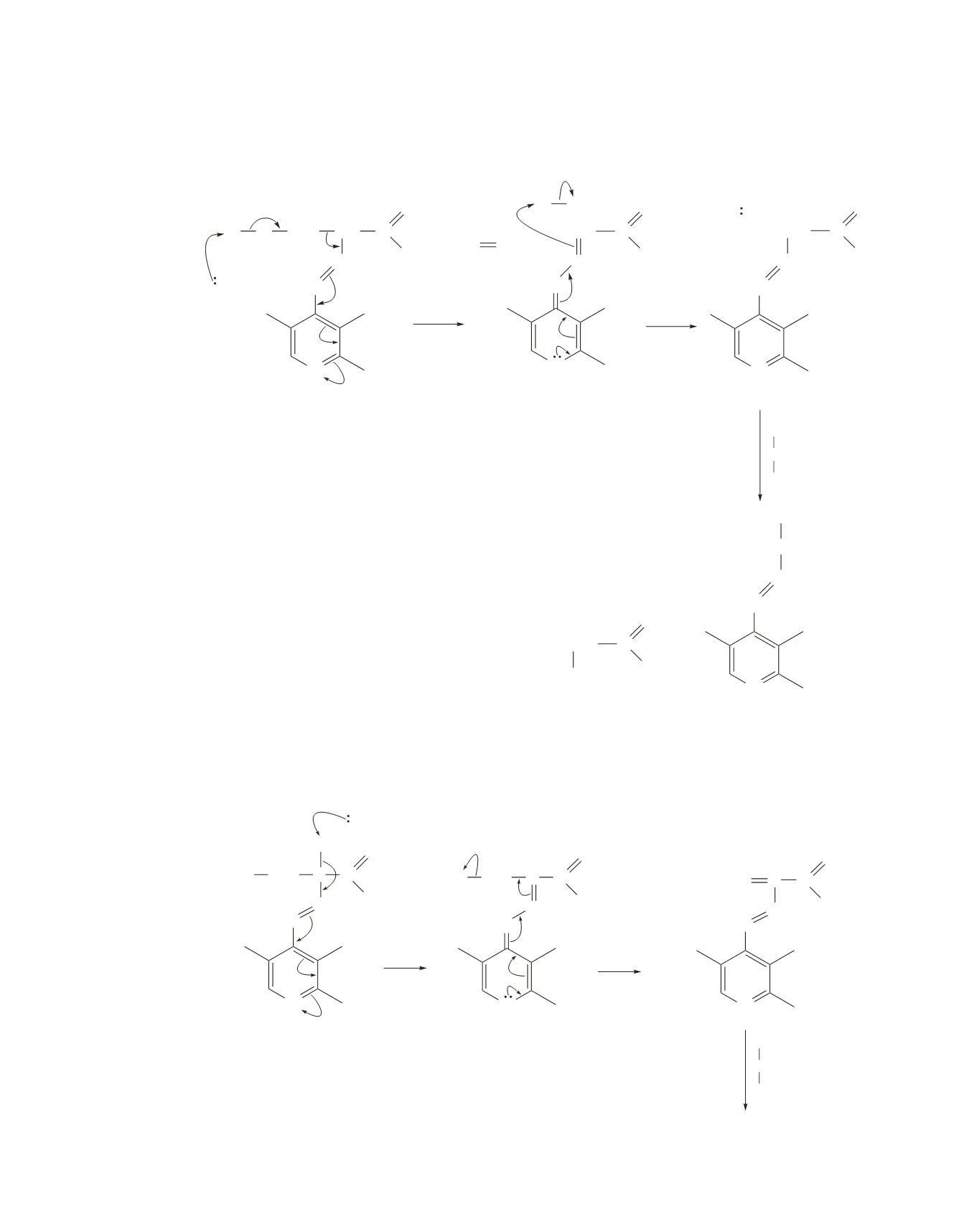
17.
For clarity, the substituents are not put on the three bonds attached to the pyridinium ring in
Problems 17 and 18.
glycine
+
(CH
2
)
4
E
CH
2
NH
2
(CH
2
)
4
NH
2
E
transimination
CH
2
H B
+
CH
2
O
N
CH C
B
HC
HC
HC
HC
N
CH CH
2
OH
_
C
O
O
_
O
O
C
_
O
O
C
_
O
O
N
N
B
+
H
N
+
H
N
+
H
N
N
H
18.
In the first step, a proton is removed from the
a
-carbon; in the next step, the leaving group is eliminated
from the
b
-carbon. Transimination produces an enamine. The enamine tautomerizes to an imine that is
hydrolyzed to the final product.
HB
+
X
_
(CH
2
)
4
N
E
(CH
2
)
4
NH
2
E
transimination
N
C CH
2
N
C CH
2
X
C
O
B
N
C CH
2
X
H
_
C
O
O
_
O
N
H
N
H
+
C
O
_
O
O
HC
HC
HC
+
H
N
















