
560 Chapter 17
Copyright © 2017 Pearson Education, Inc.
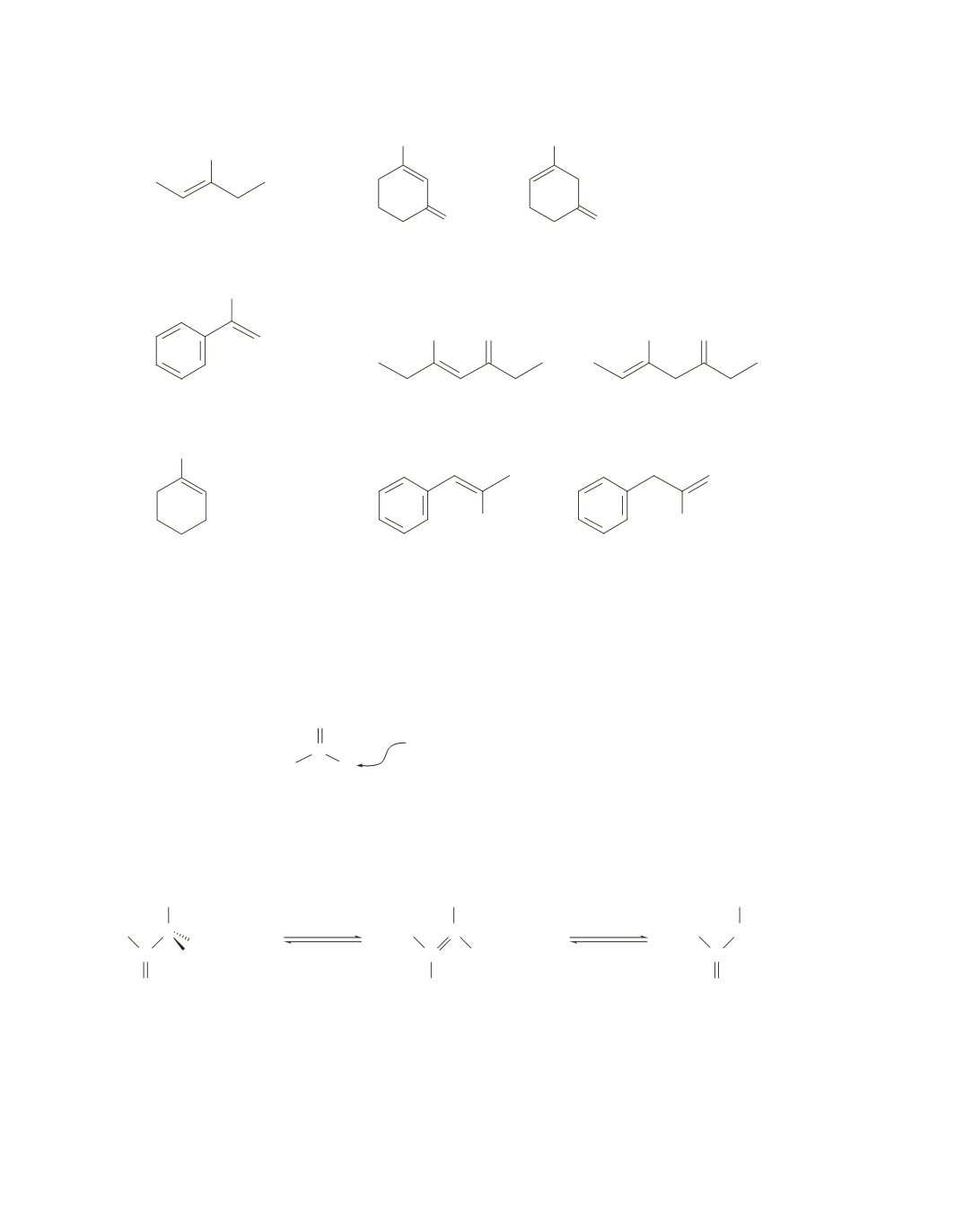
7.
d.
OH
a.
O
and
OH
O
more stable,
because the double bonds
more stable,
because the double bonds
are conjugated
more stable,
because the double bond
is conjugated with the
benzene ring
are conjugated
b.
c.
OH
e.
f.
OH
OH
OH
OH O
O
OH
OH
8.
The methyl hydrogens can be removed by a base
1
-
OD
2
and then are reprotonated by D
2
O. The aldehyde
hydrogen cannot be removed by a base because the electrons left behind if it were to be removed cannot be
delocalized.
CH
3
C
O
H
The aldehyde hydrogen is not acidic.
9.
In an acidic or basic solution, the aldehyde is in equilibrium with its enol. When the ketone enolizes, the
asymmetric center is lost. When the enol reforms the ketone, the proton can add to the
sp
2
carbon from
above or below the plane of the double bond defined by the
sp
2
carbons. As a result, equal amounts of
the
R
and
S
ketones are formed.
H
+
HO or
_
CH
3
H
H
*C
C
O
CH
2
CH
2
CH
3
CH
3
H C
C
OH
CH
2
CH
2
CH
3
H *
3
CHCH
C
O
CH
2
CH
2
CH
3
(
R
)-2-methylpentanal
R
and
S
















