
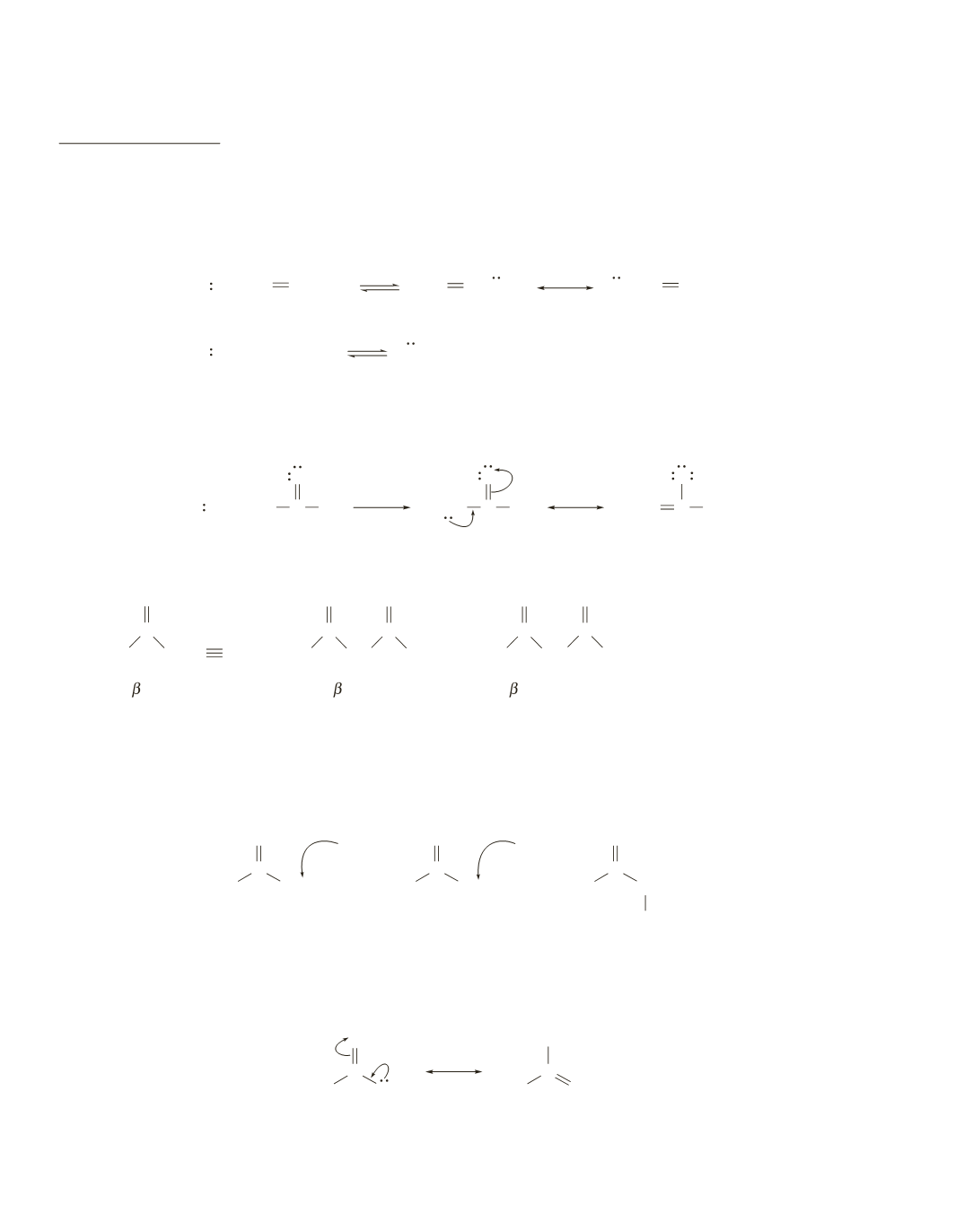
558 Chapter 17
Copyright © 2017 Pearson Education, Inc.
Solutions to Problems
1.
The electrons left behind when a base removes a proton from propene are delocalized over three carbons.
In contrast, the electrons left behind when a base removes a proton from an alkane are localized—they
belong to a single carbon. Electron delocalization stabilizes the base, and the more stable the base, the
stronger its conjugate acid. Therefore, propene is a stronger acid than an alkane.
2
3
CH CHCH
−
+
2
2
CH CHCH
HB
−
2
2
CH CH CH
3 2 3
CH CH CH
HB
+
B
−
+
+
B
−
−
CH
2
CH
2
CH
3
Propene, however, is not as acidic as the carbon acids in Table 17.1, because the electrons left behind when
a base removes a proton from these carbon acids are delocalized onto an oxygen or a nitrogen, which are
more electronegative than carbon and, therefore, are better able to accommodate the electrons.
RCH
2
C R
O
RCH C R
O
−
+
HB
RCH C R
O
−
B
−
+
2.
R
R
CH
2
C N RO CH
2
OR
CH
2
H
C
O
C
O
C
O
C
O
C
O
a -keto nitrile
a -diester
a -keto aldehyde
3.
A proton cannot be removed from the
a
-carbon of
N
-methylethanamide or ethanamide because these
compounds have a hydrogen bonded to the nitrogen and this hydrogen is more acidic than the one attached
to the
a
-carbon. Therefore, a base will remove the hydrogen attached to the nitrogen. In the case of
N
,
N
-dimethylethanamide, there is no N
¬
H proton, so a proton can be removed from the
a
-carbon.
CH
3
NCH
3
C
O
CH
3
N
,
N
-dimethylethanamide
N
-methylethanamide
ethanamide
CH
3
NHCH
3
C
O
CH
3
NH
2
C
O
The following resonance contributors show why the hydrogen attached to the nitrogen is more acidic (the
nitrogen has a partial positive charge) than the hydrogen attached to the
a
-carbon.
CH
3
NH
2
C
O
CH
3
NH
2
C
O
−
+
















