
546 Chapter 16
Copyright © 2017 Pearson Education, Inc.
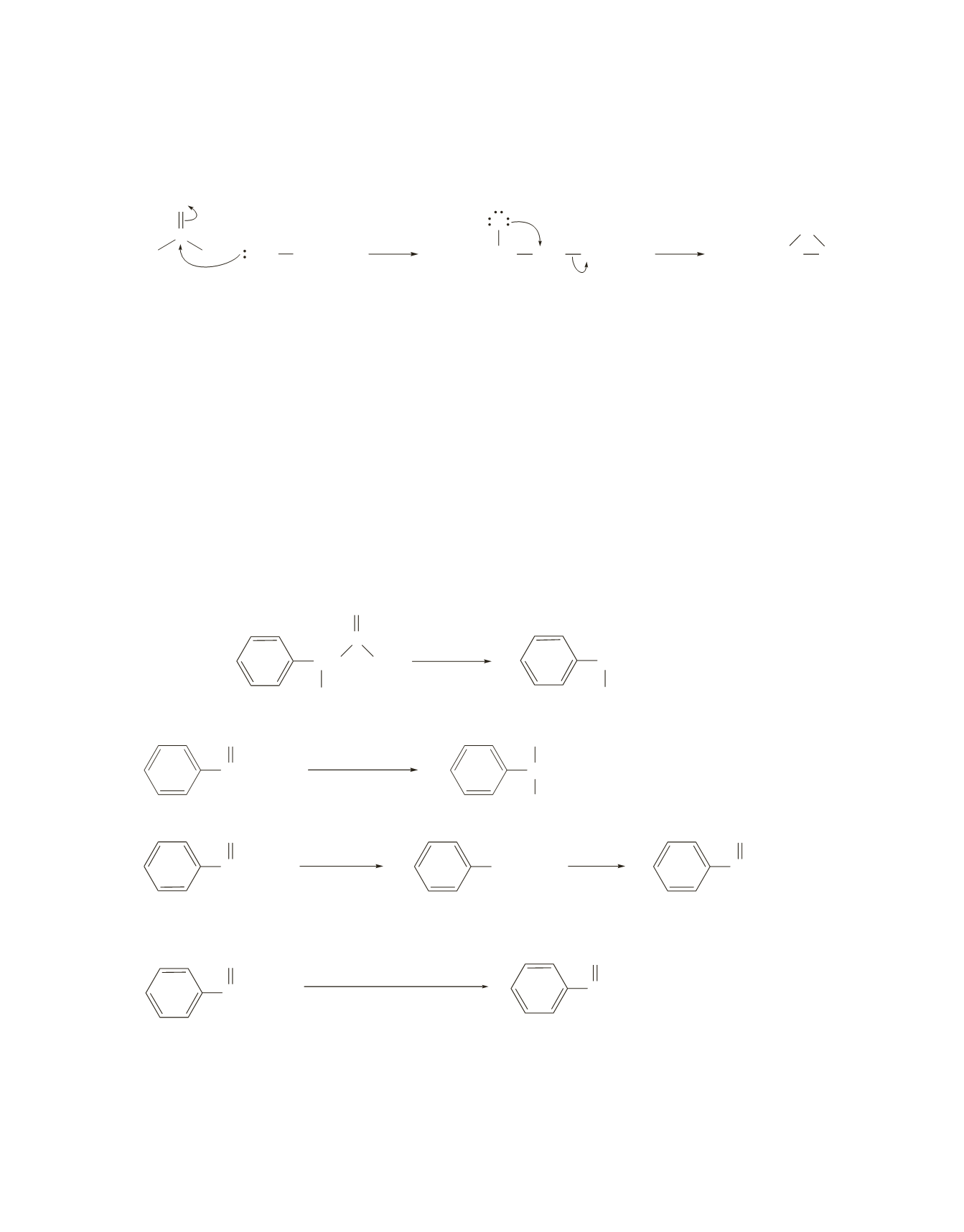
In contrast, the sulfonium ylide adds to the carbonyl group to form the intermediate shown here. The
intermediate has a nucleophile (an oxyanion) that attacks the methylenecarbon because
1
CH
3
2
2
S
is a weak
base and, therefore, a very good leaving group.
+
S(CH
3
)
2
+
O
H
CH
3
CH
2
C
−
CH
2
CH
3
CH
2
CH
S(CH
3
)
2
+
(CH
3
)
2
S
CH
2
O
CH
3
CH
2
CH CH
2
−
O
83.
The absorption bands at
1600 cm
-
1
, 1500 cm
-
1
,
and
7
3000 cm
-
1
in the IR spectrum indicate that the com-
pound has a benzene ring. The absorption band at
1720 cm
-
1
suggests that it is an aldehyde or a ketone
and the carbonyl group is not conjugated with the benzene ring. The absorption bands at
2720 cm
-
1
and
2820 cm
-
1
indicate that the compound is an aldehyde.
The broad signal at 1.8 ppm in the
1
H NMR
spectrum of the product indicates an OH group. We also see
that there are three different kinds of hydrogens (other than the OH group and the benzene ring hydrogens).
The doublet at
1.2
ppm that integrates to 3 protons indicates a methyl group adjacent to a carbon bonded
to one hydrogen. The doublet at 3.6 ppm that integrates to 2 protons is due to a methylene group bonded
to a CH group. The chemical shift of this signal suggests that the electron-withdrawing OH group is also
bonded to this methylene group.
The IR spectrum is the spectrum of 2-phenylpropanal, and the
1
H NMR
spectrum is the spectrum of
2-phenyl-1-propanol.
CH H
CH
3
1. NaBH
4
2. HCl
CHCH
2
OH
CH
3
C
O
84.
a.
COCH
3
O
1. excess CH
3
MgBr
2. HCl
CCH
3
OH
CH
3
b.
COCH
3
O
1. LiAlH
4
2. HCl
CH
2
OH
NaOCl
CH
3
COOH
0
°
C
CH
O
The synthesis can also be carried out in one step.
COCH
3
CH
1.
2.
[(CH
3
)
2
CHCH
2
]
2
AlH,
H
2
O
−
78 °C
O
O
















