
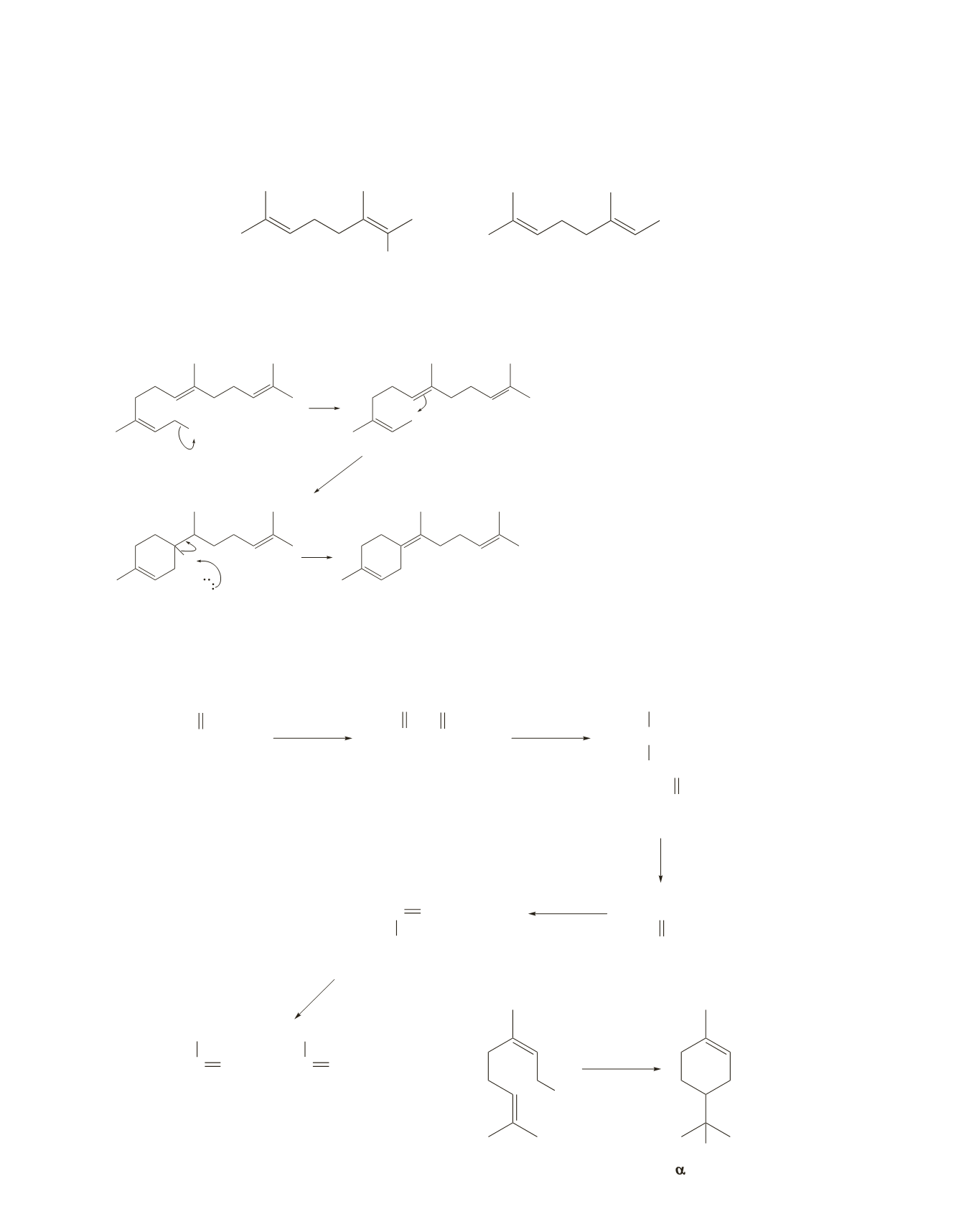
796 Chapter 25
Copyright © 2017 Pearson Education, Inc.
21.
It tells you that the reaction is an S
N
1 reaction because the fluoro-substituted carbocation is less stable than
the non-fluoro-substituted carbocation (due to the strongly electron-withdrawing fluoro substituent), so it
would form more slowly.
+
F
less stable
more stable
+
If the reaction had been an S
N
2 reaction, the fluoro-substituted compound would have reacted more rapidly
than the non-fluoro-substituted carbocation because the electron-withdrawing fluoro substituent would
make the compound more susceptible to nucleophilic attack.
22.
OPPi
H
H
2
O
+
H
3
O
+
+
+
farnesyl pyrophosphate
23.
Solved in the text.
24.
Because acetyl-CoA is converted into malonyl-CoA (see Section 25.8), mevalonyl pyrophosphate will contain
three labeled carbons, which means that the
a
-terpineol (juniper oil) will contain six labeled carbons.
CH
3
CSCoA
14
O
−
OCCH
2
CSCoA
CH
3
CCH
2
CH
2
OPP
i
OH
CH
2
CO
−
O
mevalonyl pyrophosphate
CH
3
CCH
2
CH
2
OPP
i
OPP
i
CH
2
CH
3
C CHCH
2
CH
3
dimethylallyl pyrophosphate
CH
3
C CHCH
2
CH
2
C CHCH
2
OPP
i
CH
3
CH
3
geranyl pyrophosphate
OPP
i
14
14
14
14
14
14
=
14
14
14
14
14
14
OH
geranyl pyrophosphate
-terpineol
14
O
14 14
14
14 14
14
14
14
14
14
14
14
14
14
isopentenyl pyrophosphate
O
















