
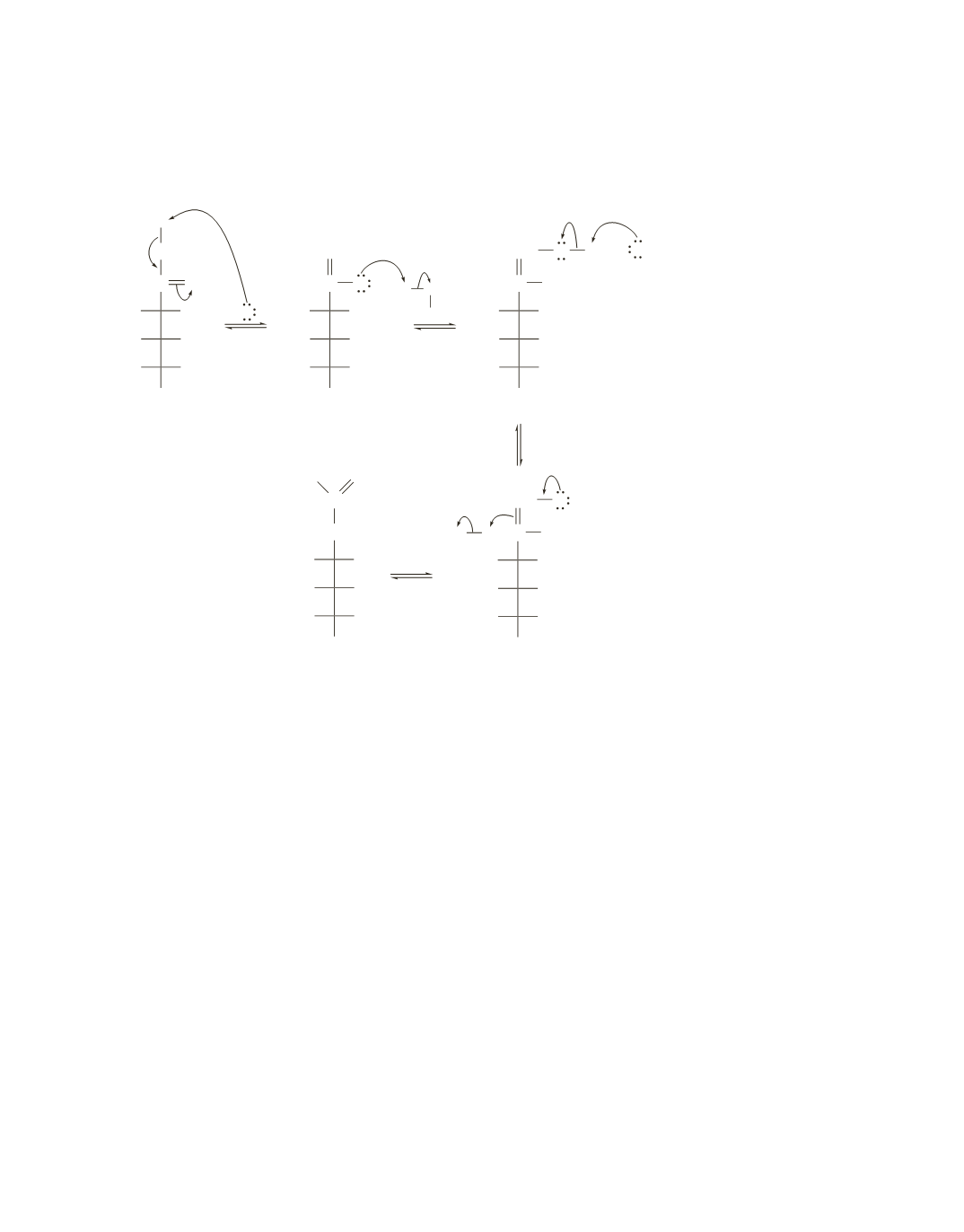
678 Chapter 20
Copyright © 2017 Pearson Education, Inc.
10.
Removal of an
a
-hydrogen creates an enol that can enolize back to the ketone (using the OH at C-2) or can
enolize to an aldehyde (using the OH at C-1). The aldehyde has a new asymmetric center (indicated by an *);
one of the epimers is d-glucose, and the other is d-mannose.
CHOH
C O
HO H
H OH
H OH
CH
2
OH
CH
C OH
HO H
H OH
H OH
CH
2
OH
D
-fructose
H
O H OH
−
HO
H
2
O
−
−
CHOH
HO
HO
H
H OH
H OH
CH
2
OH
D
-glucose
D
-mannose
−
*
C
H O
H
H
O
CHOH
C O
HO H
H OH
H OH
CH
2
OH
HO H
CH
C OH
HO H
H OH
H OH
CH
2
OH
O
−
















